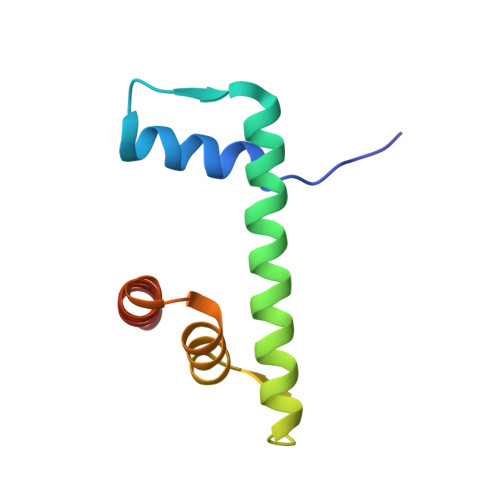
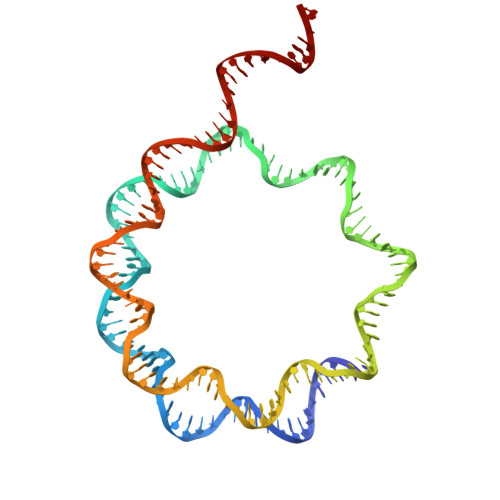
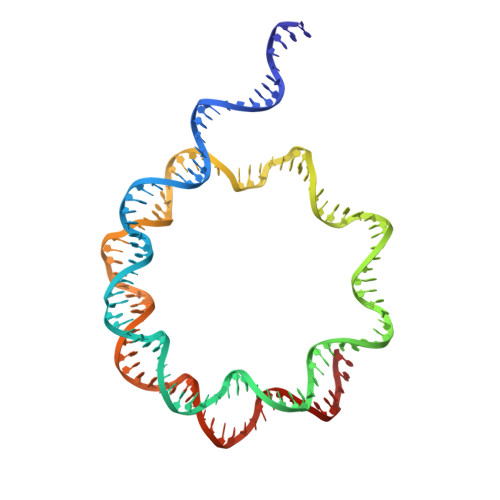
Recognition and remodelling of nucleosomes and hexasomes by the human INO80 complex.
Aggarwal, P., Sharma, M., Woike, S., Kunert, F., Brem, A., Moldt, M., Hopfner, K.P.(2026) Nucleic Acids Res 54
- PubMed: 41775336 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1093/nar/gkag138
- Primary Citation Related Structures:
9GE4, 9GEL, 9GF6, 9GFM - PubMed Abstract:
The ATP-dependent INO80 chromatin remodeller slides and repositions nucleosomes to shape and maintain chromatin around gene regulatory elements and replication origins. Recent work uncovered capabilities of yeast and fungal INO80 to bind and slide hexasomes, but whether this is a universal feature is unknown. Here, we show that human INO80 also slides hexasomes as efficiently as H2A and H2A.Z nucleosomes. By determining a variety of structures of human INO80 bound to canonical and H2A.Z nucleosomes as well as hexasomes, we reveal a predominantly topological sensing of nucleosomal species with at least three positions depending on entry DNA unwrapping. INO80 spin-rotates around the nucleosomal core particle as a function of entry DNA unwrapping. Different degrees of unwrapped entry DNA lead to two different nucleosomal and one hexasomal locations of INO80, determined by binding of the Snf2 ATPase to entry point of extranucleosomal DNA at the nucleosome/hexasome core. Acidic patch binding by the INO80 subunit IES2 can differentiate between (sub)nucleosomal species, is important for nucleosome but not hexasome sliding, and may sense unwrapped exit DNA. These findings provide structural and mechanistic insights into how human INO80 remodels diverse chromatin substrates in a topology driven manner.
- Gene Center, Department of Biochemistry, Ludwig-Maximilians-Universität München, Feodor-Lynen-Str. 25, 81377 Munich, Germany.
Organizational Affiliation: