CryoEM Structure of Glutamate transporter homologue GltPh mutant P206R in complex with L-Aspartate and Sodium ions in Salipro
Horn, G., Overa, C., Fu, L., Urbansky, K., Madej, M.G., Ziegler, C.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Glutamate transporter homolog | 425 | Pyrococcus horikoshii | Mutation(s): 1 Gene Names: PH1295 | 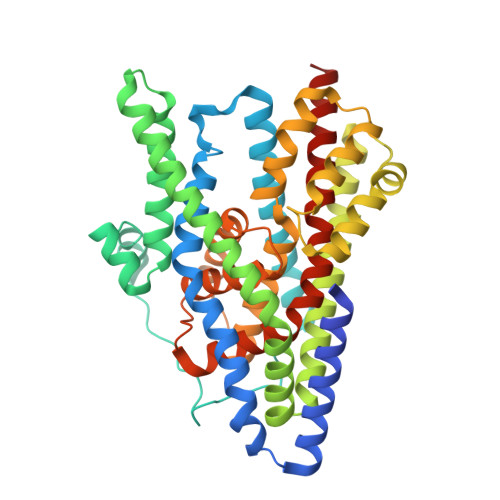 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | O59010 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 6 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| D3D (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | J [auth A], Q [auth B], R [auth C] | (19S,22R,25R)-22,25,26-trihydroxy-16,22-dioxo-17,21,23-trioxa-22lambda~5~-phosphahexacosan-19-yl (9E)-octadec-9-enoate C40 H77 O10 P PAZGBAOHGQRCBP-UCZVXENKSA-N |  | ||
| PG8 (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | F [auth A], M [auth B], U [auth C] | 1,2-DIOCTANOYL-SN-GLYCERO-3-[PHOSPHO-RAC-(1-GLYCEROL) C22 H42 O10 P BQEXNLVNNRZNEI-WOJBJXKFSA-M |  | ||
| PGM (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | G [auth A], N [auth B], V [auth C] | 1-MYRISTOYL-2-HYDROXY-SN-GLYCERO-3-[PHOSPHO-RAC-(1-GLYCEROL)] C22 H44 O9 P BVJSKAUUFXBDOB-SFTDATJTSA-M |  | ||
| ASP (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | D [auth A], K [auth B], S [auth C] | ASPARTIC ACID C4 H7 N O4 CKLJMWTZIZZHCS-REOHCLBHSA-N |  | ||
| CL Download:Ideal Coordinates CCD File | H [auth A] I [auth A] O [auth B] P [auth B] W [auth C] | CHLORIDE ION Cl VEXZGXHMUGYJMC-UHFFFAOYSA-M |  | ||
| NA Download:Ideal Coordinates CCD File | E [auth A], L [auth B], T [auth C] | SODIUM ION Na FKNQFGJONOIPTF-UHFFFAOYSA-N |  | ||
| Task | Software Package | Version |
|---|---|---|
| RECONSTRUCTION | cryoSPARC | |
| MODEL REFINEMENT | REFMAC | 5 |
| Funding Organization | Location | Grant Number |
|---|---|---|
| German Research Foundation (DFG) | Germany | 2518 |