The structure of NiaR from Thermotoga maritima bound to nicotinic acid
Glasfeld, A., Cheng, D.W.C., Li, Y.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Probable transcription repressor NiaR | 175 | Thermotoga maritima | Mutation(s): 0 Gene Names: niaR, TM_1602 | 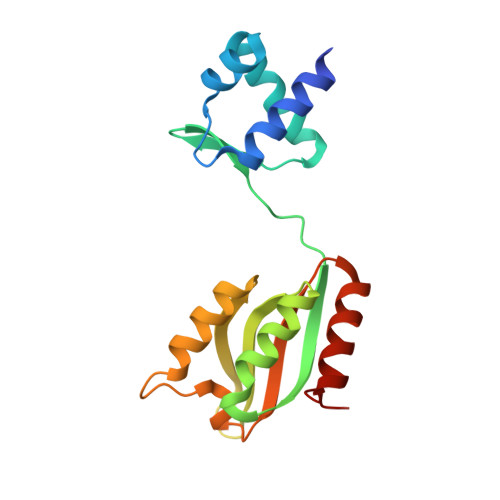 | |
UniProt | |||||
Find proteins for Q9X1T8 (Thermotoga maritima (strain ATCC 43589 / DSM 3109 / JCM 10099 / NBRC 100826 / MSB8)) Explore Q9X1T8 Go to UniProtKB: Q9X1T8 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9X1T8 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| NIO (Subject of Investigation/LOI) Query on NIO | C [auth A] | NICOTINIC ACID C6 H5 N O2 PVNIIMVLHYAWGP-UHFFFAOYSA-N |  | ||
| PRO Query on PRO | D [auth A] | PROLINE C5 H9 N O2 ONIBWKKTOPOVIA-BYPYZUCNSA-N |  | ||
| FE2 (Subject of Investigation/LOI) Query on FE2 | B [auth A] | FE (II) ION Fe CWYNVVGOOAEACU-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 83.243 | α = 90 |
| b = 83.243 | β = 90 |
| c = 66.838 | γ = 120 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| XDS | data reduction |
| Aimless | data scaling |
| PHASER | phasing |
| Coot | model building |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Not funded | -- |