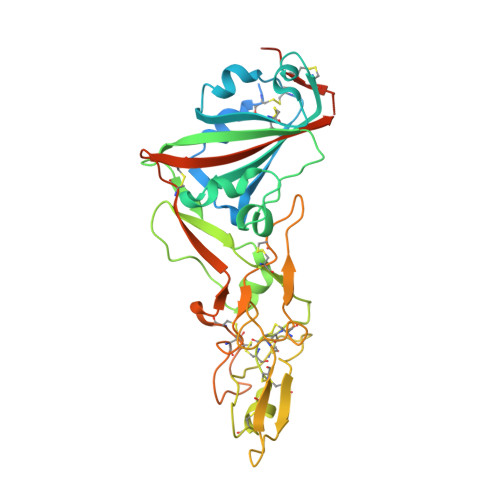
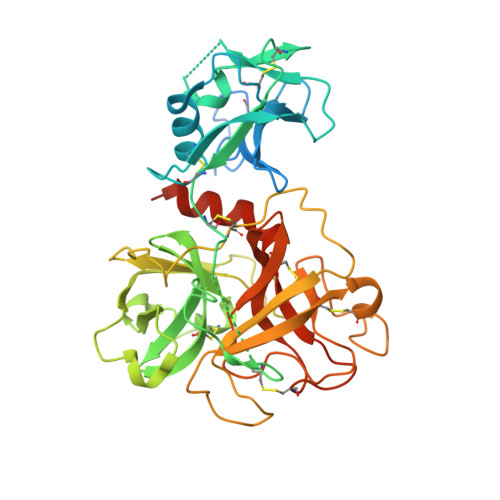
Human coronavirus HKU1 recognition of the TMPRSS2 host receptor.
McCallum, M., Park, Y.J., Stewart, C., Sprouse, K.R., Addetia, A., Brown, J., Tortorici, M.A., Gibson, C., Wong, E., Ieven, M., Telenti, A., Veesler, D.(2024) Cell 187: 4231
- PubMed: 38964328 Search on PubMed
- DOI: https://doi.org/10.1016/j.cell.2024.06.006
- Primary Citation Related Structures:
8VGT - PubMed Abstract:
The human coronavirus HKU1 spike (S) glycoprotein engages host cell surface sialoglycans and transmembrane protease serine 2 (TMPRSS2) to initiate infection. The molecular basis of HKU1 binding to TMPRSS2 and determinants of host receptor tropism remain elusive. We designed an active human TMPRSS2 construct enabling high-yield recombinant production in human cells of this key therapeutic target. We determined a cryo-electron microscopy structure of the HKU1 RBD bound to human TMPRSS2, providing a blueprint of the interactions supporting viral entry and explaining the specificity for TMPRSS2 among orthologous proteases. We identified TMPRSS2 orthologs from five mammalian orders promoting HKU1 S-mediated entry into cells along with key residues governing host receptor usage. Our data show that the TMPRSS2 binding motif is a site of vulnerability to neutralizing antibodies and suggest that HKU1 uses S conformational masking and glycan shielding to balance immune evasion and receptor engagement.
- Department of Biochemistry, University of Washington, Seattle, WA, USA.
Organizational Affiliation: