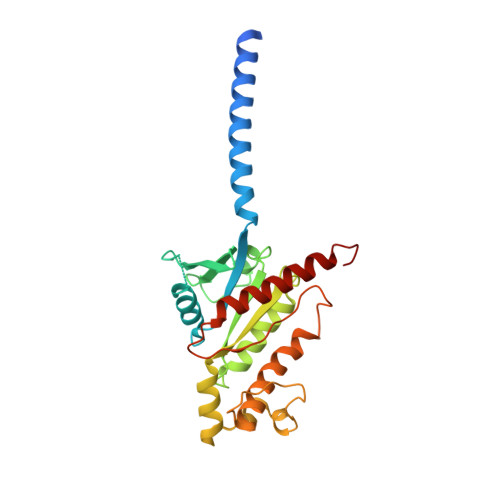
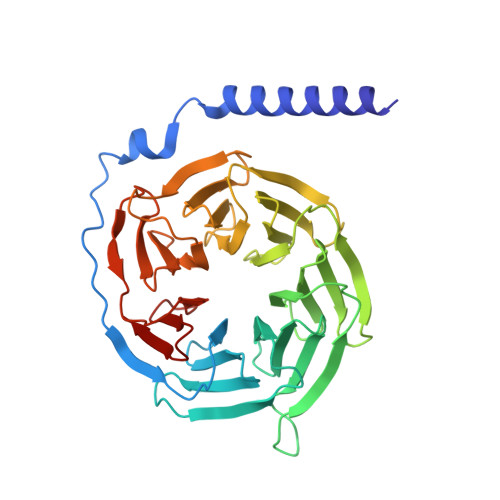
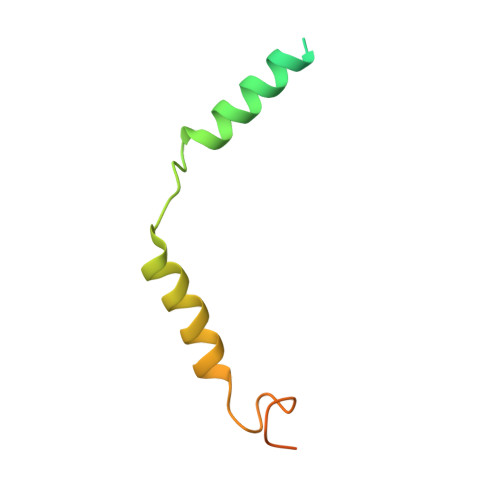
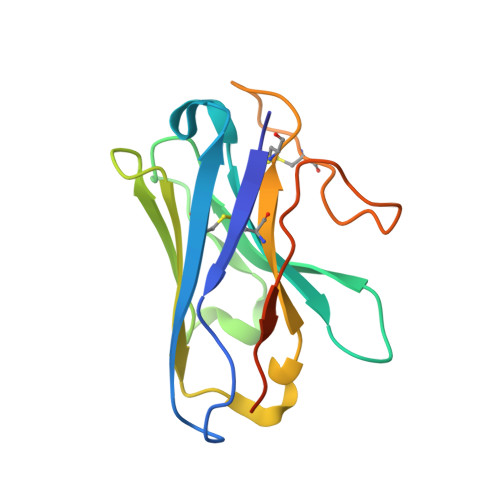
Fenofibrate Recognition and G q Protein Coupling Mechanisms of the Human Cannabinoid Receptor CB1.
Wang, T., Tang, W., Zhao, Z., Zhao, R., Lv, Z., Guo, X., Gu, Q., Liu, B., Lv, H., Chen, J., Zhang, K., Li, F., Wang, J.(2024) Adv Sci (Weinh) 11: e2306311-e2306311
- PubMed: 38298116 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1002/advs.202306311
- Primary Citation Related Structures:
8K8J - PubMed Abstract:
The G-protein-coupled human cannabinoid receptor 1 (CB1) is a promising therapeutic target for pain management, inflammation, obesity, and substance abuse disorders. The structures of CB1-G i complexes in synthetic agonist-bound forms have been resolved to date. However, the commercial drug recognition and G q coupling mechanisms of CB1 remain elusive. Herein, the cryo-electron microscopy (cryo-EM) structure of CB1-G q complex, in fenofibrate-bound form, at near-atomic resolution, is reported. The structure elucidates the delicate mechanisms of the precise fenofibrate recognition and G q protein coupling by CB1 and will facilitate future drug discovery and design.
- CAS Key Laboratory of Quantitative Engineering Biology, Institute of Synthetic Biology, Shenzhen Institute of Advanced Technology, Chinese Academy of Sciences, Shenzhen, 518055, China.
Organizational Affiliation: