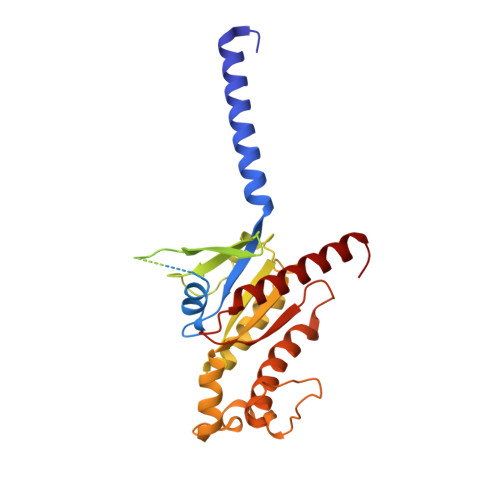
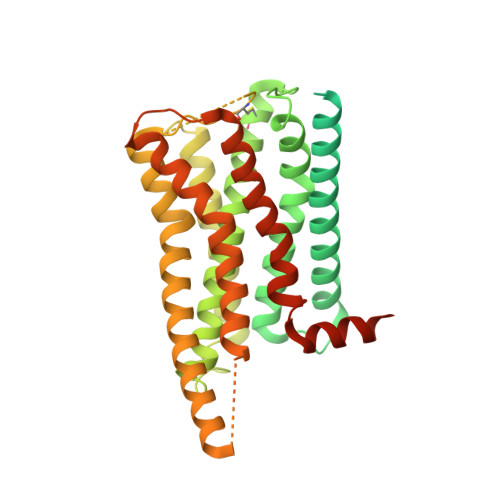
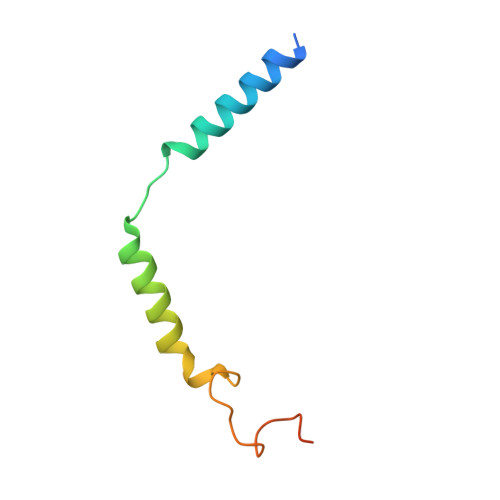
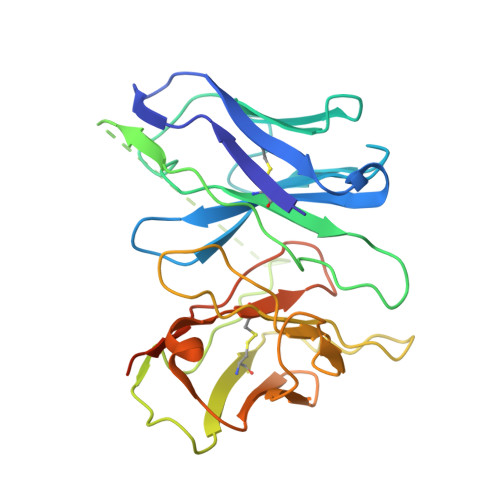
Ligand recognition and G-protein coupling of trace amine receptor TAAR1.
Xu, Z., Guo, L., Yu, J., Shen, S., Wu, C., Zhang, W., Zhao, C., Deng, Y., Tian, X., Feng, Y., Hou, H., Su, L., Wang, H., Guo, S., Wang, H., Wang, K., Chen, P., Zhao, J., Zhang, X., Yong, X., Cheng, L., Liu, L., Yang, S., Yang, F., Wang, X., Yu, X., Xu, Y., Sun, J.P., Yan, W., Shao, Z.(2023) Nature 624: 672-681
- PubMed: 37935376 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41586-023-06804-z
- Primary Citation Related Structures:
8JLJ, 8JLK, 8JLN, 8JLO, 8JLP, 8JLQ, 8JLR, 8JSO, 8JSP - PubMed Abstract:
Trace-amine-associated receptors (TAARs), a group of biogenic amine receptors, have essential roles in neurological and metabolic homeostasis 1 . They recognize diverse endogenous trace amines and subsequently activate a range of G-protein-subtype signalling pathways 2,3 . Notably, TAAR1 has emerged as a promising therapeutic target for treating psychiatric disorders 4,5 . However, the molecular mechanisms underlying its ability to recognize different ligands remain largely unclear. Here we present nine cryo-electron microscopy structures, with eight showing human and mouse TAAR1 in a complex with an array of ligands, including the endogenous 3-iodothyronamine, two antipsychotic agents, the psychoactive drug amphetamine and two identified catecholamine agonists, and one showing 5-HT 1A R in a complex with an antipsychotic agent. These structures reveal a rigid consensus binding motif in TAAR1 that binds to endogenous trace amine stimuli and two extended binding pockets that accommodate diverse chemotypes. Combined with mutational analysis, functional assays and molecular dynamic simulations, we elucidate the structural basis of drug polypharmacology and identify the species-specific differences between human and mouse TAAR1. Our study provides insights into the mechanism of ligand recognition and G-protein selectivity by TAAR1, which may help in the discovery of ligands or therapeutic strategies for neurological and metabolic disorders.
- Division of Nephrology and Kidney Research Institute, State Key Laboratory of Biotherapy, West China Hospital, Sichuan University, Chengdu, China.
Organizational Affiliation: