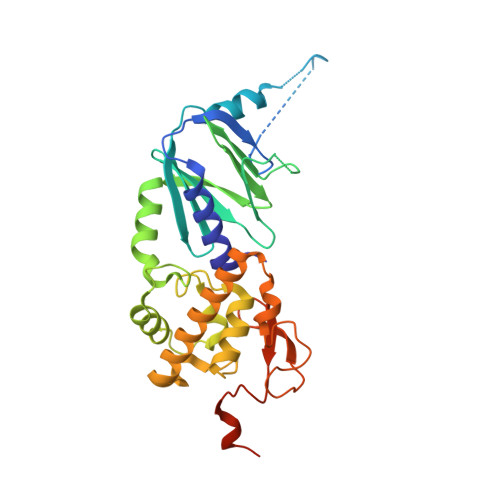
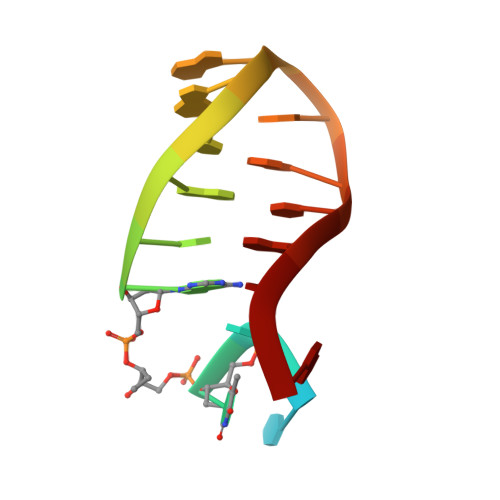
Model of abasic site DNA cross-link repair; from the architecture of NEIL3 DNA binding domains to the X-structure model.
Huskova, A., Dinesh, D.C., Srb, P., Boura, E., Veverka, V., Silhan, J.(2022) Nucleic Acids Res 50: 10436-10448
- PubMed: 36155818 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1093/nar/gkac793
- Primary Citation Related Structures:
7OMK, 7Z5A - PubMed Abstract:
Covalent DNA interstrand crosslinks are toxic DNA damage lesions that block the replication machinery that can cause a genomic instability. Ubiquitous abasic DNA sites are particularly susceptible to spontaneous cross-linking with a base from the opposite DNA strand. Detection of a crosslink induces the DNA helicase ubiquitination that recruits NEIL3, a DNA glycosylase responsible for the lesion removal. NEIL3 utilizes several zinc finger domains indispensable for its catalytic NEI domain repairing activity. They recruit NEIL3 to the repair site and bind the single-stranded DNA. However, the molecular mechanism underlying their roles in the repair process is unknown. Here, we report the structure of the tandem zinc-finger GRF domain of NEIL3 and reveal the molecular details of its interaction with DNA. Our biochemical data indicate the preferential binding of the GRF domain to the replication fork. In addition, we obtained a structure for the catalytic NEI domain in complex with the DNA reaction intermediate that allowed us to construct and validate a model for the interplay between the NEI and GRF domains in the recognition of an interstrand cross-link. Our results suggest a mechanism for recognition of the DNA replication X-structure by NEIL3, a key step in the interstrand cross-link repair.
- Institute of Organic Chemistry and Biochemistry of the Czech Academy of Sciences, Flemingovo namesti 2, 166 10 Prague, Czech Republic.
Organizational Affiliation: