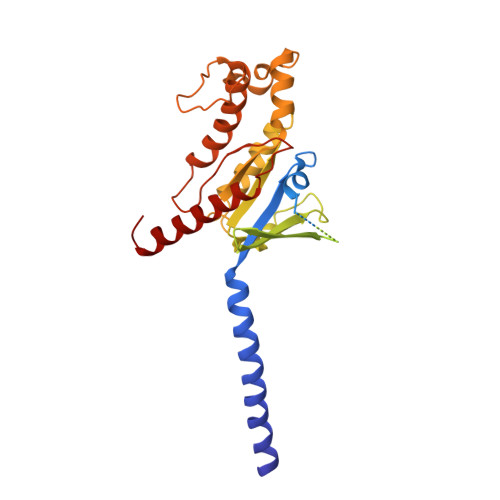
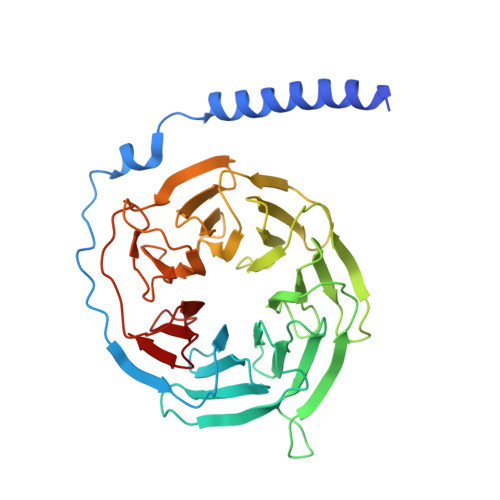
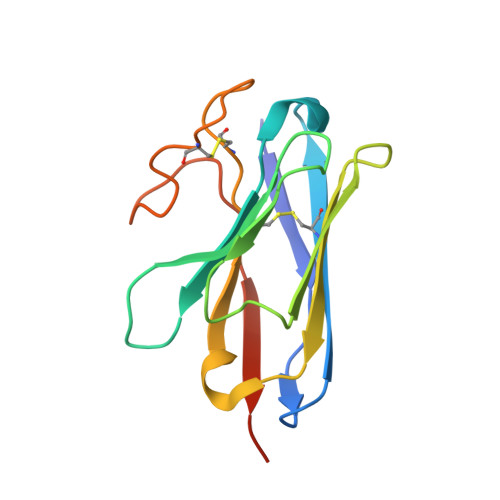
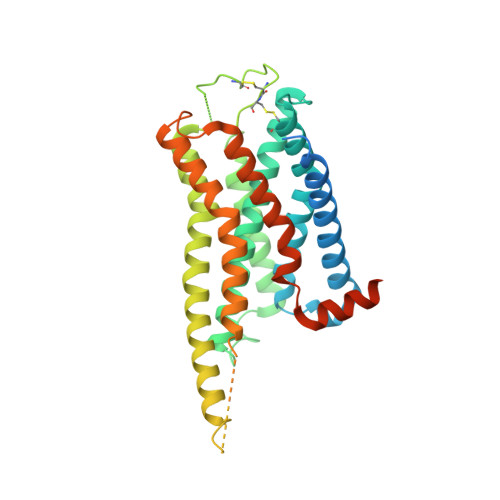
Cryo-EM structures of the beta 3 adrenergic receptor bound to solabegron and isoproterenol.
Nureki, I., Kobayashi, K., Tanaka, T., Demura, K., Inoue, A., Shihoya, W., Nureki, O.(2022) Biochem Biophys Res Commun 611: 158-164
- PubMed: 35489202 Search on PubMed
- DOI: https://doi.org/10.1016/j.bbrc.2022.04.065
- Primary Citation Related Structures:
7XJI - PubMed Abstract:
The β 3 -adrenergic receptor (β 3 AR) is the most essential drug target for overactive bladder and has therapeutic potentials for the treatments of type 2 diabetes and obesity. Here, we report the cryo-electron microscopy structures of the β 3 AR-G s signaling complexes with the selective agonist, solabegron and the nonselective agonist, isoproterenol. Comparison of the isoproterenol-, mirabegron-, and solabegron-bound β 3 AR structures revealed that the extracellular loop 2 changes its conformation depending on the bound agonist and plays an essential role in solabegron binding. Moreover, β 3 AR has an intrinsically narrow exosite, regardless of the agonist type. This structural feature clearly explains why β 3 AR prefers mirabegron and solabegron, as the narrow exosite is suitable for binding with agonists with elongated shapes. Our study deepens the understanding of the binding characteristics of β 3 AR agonists and may pave the way for developing β 3 AR-selective drugs.
- Department of Biological Sciences, Graduate School of Science, The University of Tokyo, 7-3-1 Hongo, Bunkyo-ku, Tokyo, 113-0033, Japan.
Organizational Affiliation: