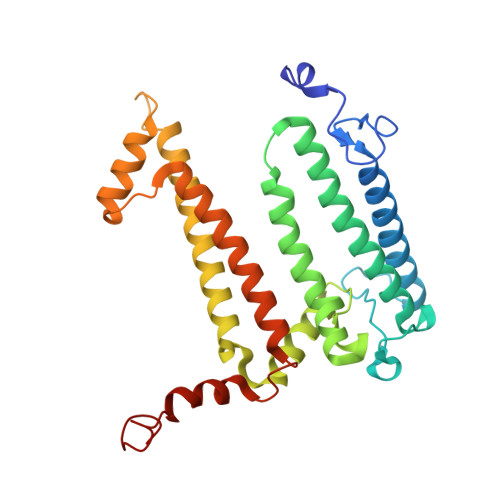
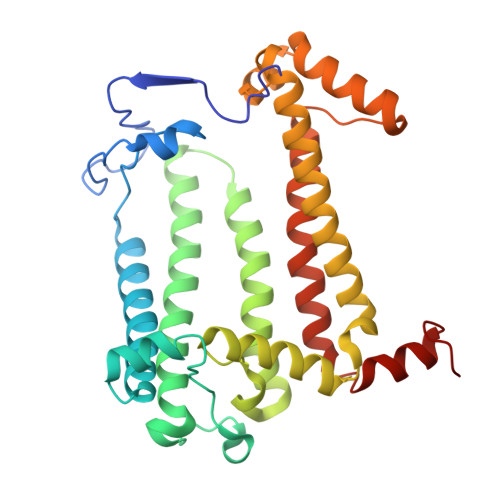
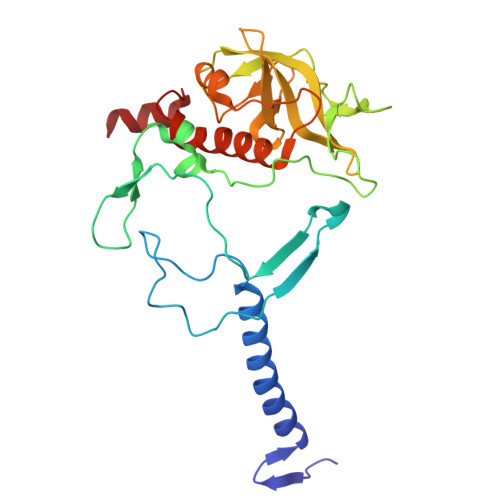
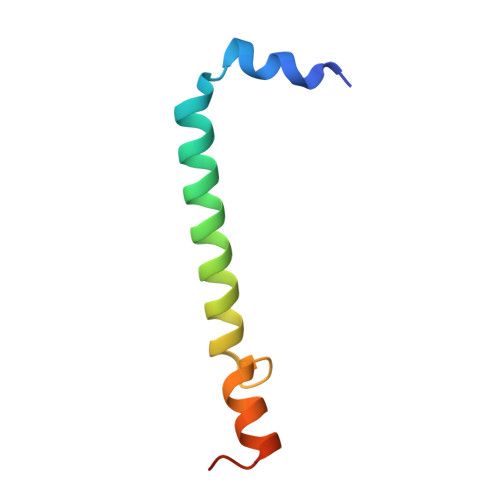
Structural basis for the assembly and quinone transport mechanisms of the dimeric photosynthetic RC-LH1 supercomplex.
Cao, P., Bracun, L., Yamagata, A., Christianson, B.M., Negami, T., Zou, B., Terada, T., Canniffe, D.P., Shirouzu, M., Li, M., Liu, L.N.(2022) Nat Commun 13: 1977-1977
- PubMed: 35418573 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-022-29563-3
- Primary Citation Related Structures:
7VA9, 7VB9, 7VNM, 7VNY, 7VOR, 7VOT, 7VOY - PubMed Abstract:
The reaction center (RC) and light-harvesting complex 1 (LH1) form a RC-LH1 core supercomplex that is vital for the primary reactions of photosynthesis in purple phototrophic bacteria. Some species possess the dimeric RC-LH1 complex with a transmembrane polypeptide PufX, representing the largest photosynthetic complex in anoxygenic phototrophs. However, the details of the architecture and assembly mechanism of the RC-LH1 dimer are unclear. Here we report seven cryo-electron microscopy (cryo-EM) structures of RC-LH1 supercomplexes from Rhodobacter sphaeroides. Our structures reveal that two PufX polypeptides are positioned in the center of the S-shaped RC-LH1 dimer, interlocking association between the components and mediating RC-LH1 dimerization. Moreover, we identify another transmembrane peptide, designated PufY, which is located between the RC and LH1 subunits near the LH1 opening. PufY binds a quinone molecule and prevents LH1 subunits from completely encircling the RC, creating a channel for quinone/quinol exchange. Genetic mutagenesis, cryo-EM structures, and computational simulations provide a mechanistic understanding of the assembly and electron transport pathways of the RC-LH1 dimer and elucidate the roles of individual components in ensuring the structural and functional integrity of the photosynthetic supercomplex.
- National Laboratory of Biomacromolecules, CAS Center for Excellence in Biomacromolecules, Institute of Biophysics, Chinese Academy of Sciences, Beijing, 100101, China.
Organizational Affiliation: