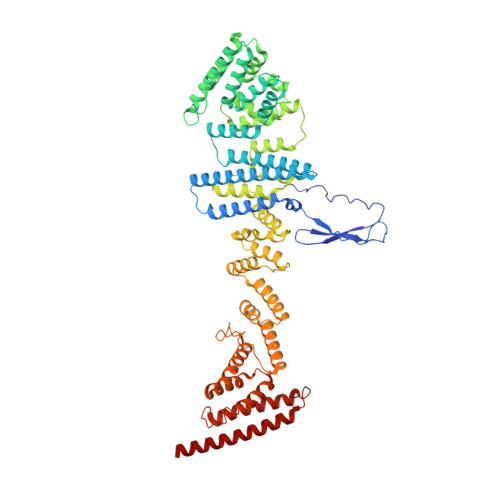
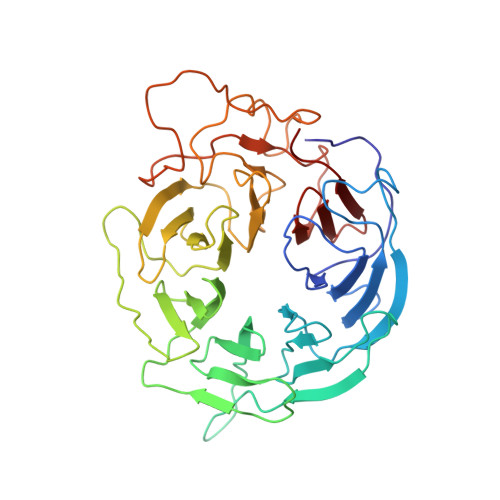
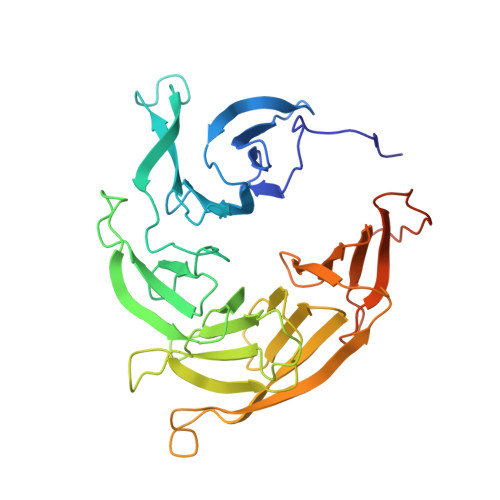
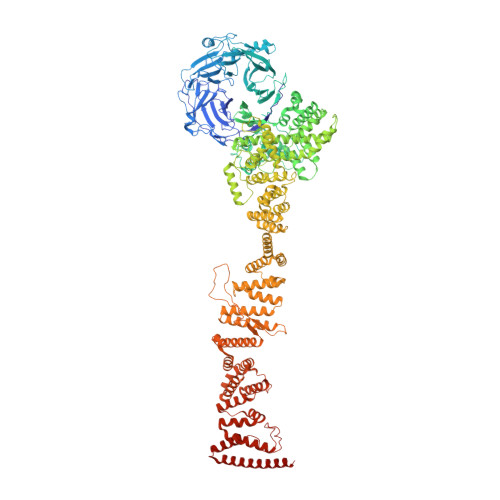
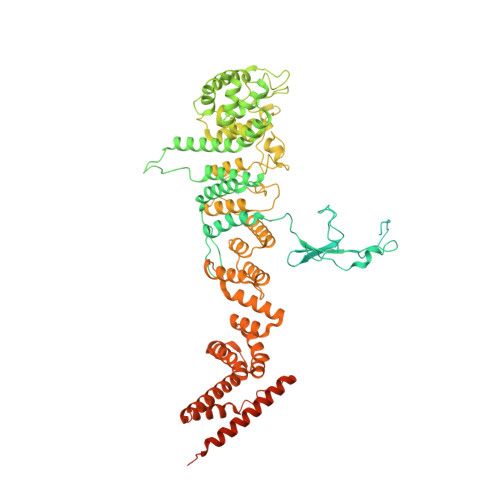
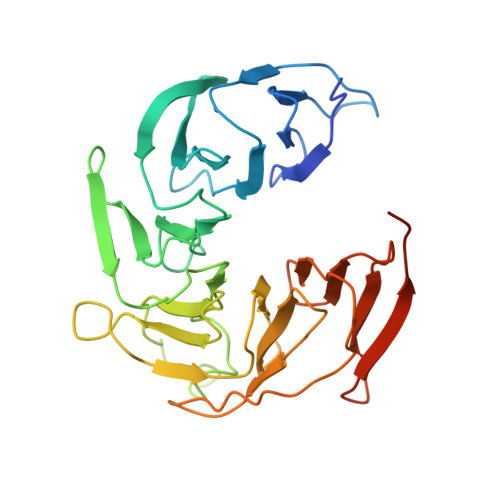
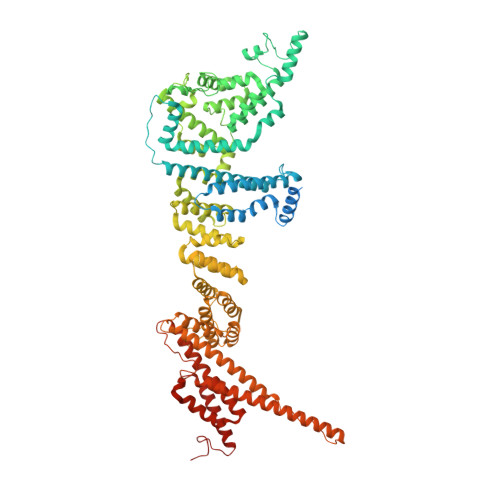
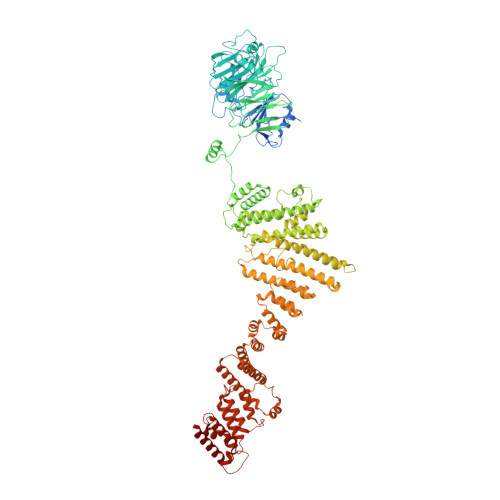
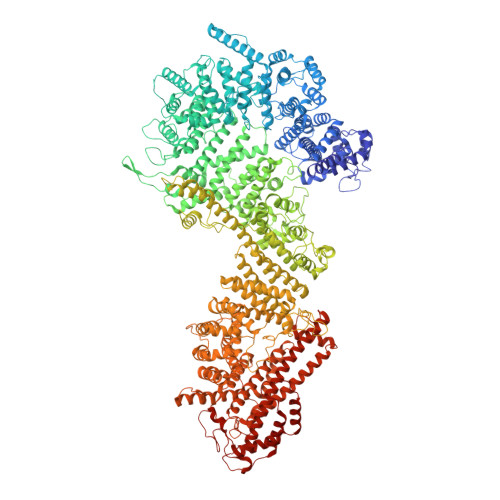
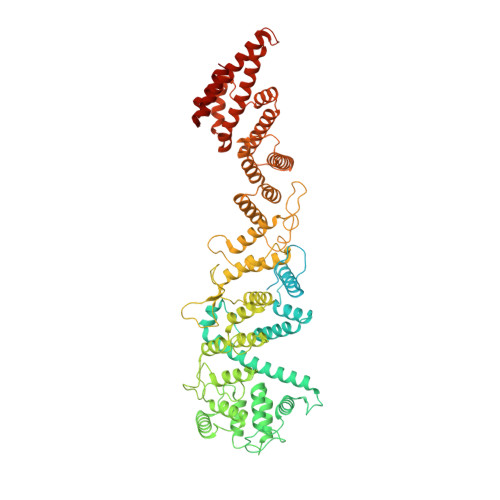
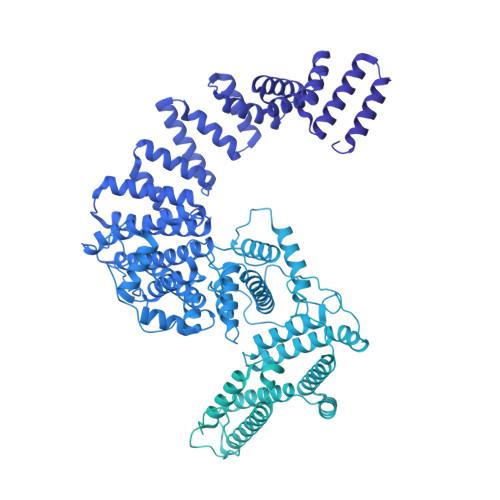
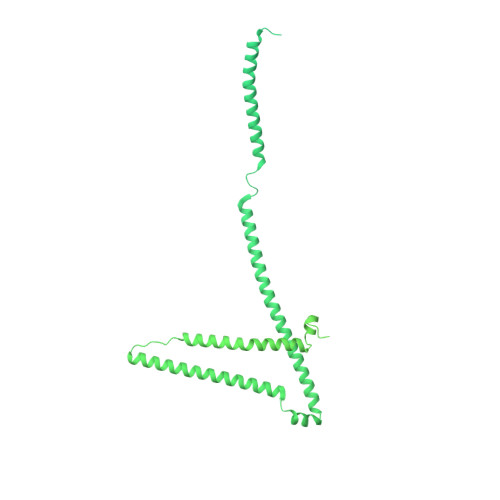
8 angstrom structure of the outer rings of the Xenopus laevis nuclear pore complex obtained by cryo-EM and AI.
Tai, L., Zhu, Y., Ren, H., Huang, X., Zhang, C., Sun, F.(2022) Protein Cell 13: 760-777
- PubMed: 35015240 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1007/s13238-021-00895-y
- Primary Citation Related Structures:
7VCI, 7VOP - PubMed Abstract:
The nuclear pore complex (NPC), one of the largest protein complexes in eukaryotes, serves as a physical gate to regulate nucleocytoplasmic transport. Here, we determined the 8 Å resolution cryo-electron microscopic (cryo-EM) structure of the outer rings containing nuclear ring (NR) and cytoplasmic ring (CR) from the Xenopus laevis NPC, with local resolutions reaching 4.9 Å. With the aid of AlphaFold2, we managed to build a pseudoatomic model of the outer rings, including the Y complexes and flanking components. In this most comprehensive and accurate model of outer rings to date, the almost complete Y complex structure exhibits much tighter interaction in the hub region. In addition to two copies of Y complexes, each asymmetric subunit in CR contains five copies of Nup358, two copies of the Nup214 complex, two copies of Nup205 and one copy of newly identified Nup93, while that in NR contains one copy of Nup205, one copy of ELYS and one copy of Nup93. These in-depth structural features represent a great advance in understanding the assembly of NPCs.
- National Key Laboratory of Biomacromolecules, Institute of Biophysics, CAS Center for Excellence in Biomacromolecules, Chinese Academy of Sciences, Beijing, 100101, China.
Organizational Affiliation: