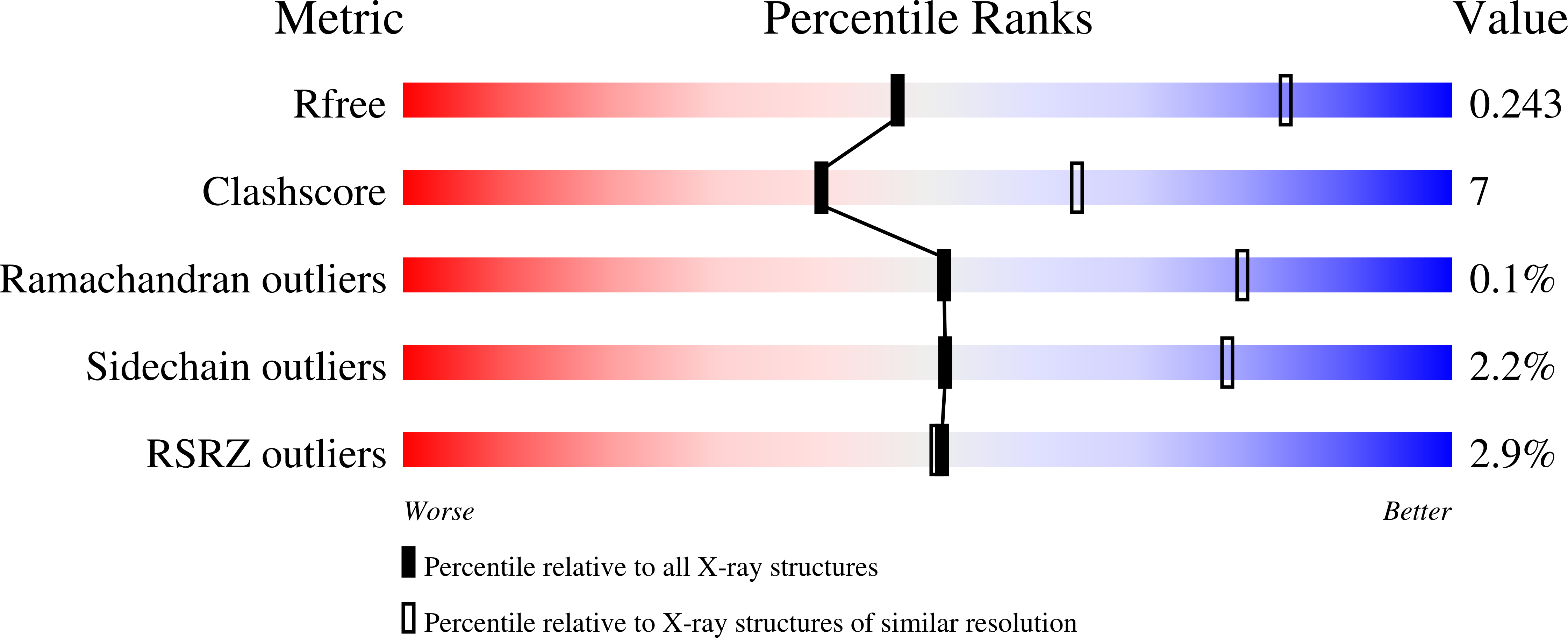
Structural and functional insights into aldosterone synthase interaction with its redox partner protein adrenodoxin.
Brixius-Anderko, S., Scott, E.E.(2021) J Biol Chem 296: 100794-100794
- PubMed: 34015331
- DOI: https://doi.org/10.1016/j.jbc.2021.100794
- Primary Citation of Related Structures:
7M8I - PubMed Abstract:
Aldosterone is the major mineralocorticoid in the human body controlling blood pressure and salt homeostasis. Overproduction of aldosterone leads to primary aldosteronism, which is the most common form of secondary hypertension with limited treatment options. Production of aldosterone by cytochrome P450 11B2 (CYP11B2, aldosterone synthase) requires two reduction events with the electrons delivered by the iron/sulfur protein adrenodoxin. Very limited information is available about the structural and functional basis of adrenodoxin/CYP11B2 interaction, which impedes the development of new treatment options for primary aldosteronism. A systematic study was carried out to determine if adrenodoxin interaction with CYP11B2 might also have an allosteric component in addition to electron transfer. Indeed, local increases in adrenodoxin concentration promote binding of the substrate 11-deoxycorticosterone and the inhibitor osilodrostat (LCI699) in the active site-over 17 Å away-as well as enhance the inhibitory effect of this latter drug. The CYP11B2 structure in complex with adrenodoxin identified specific residues at the protein-protein interface interacting via five salt bridges and four hydrogen bonds. Comparisons with cholesterol-metabolizing CYP11A1 and cortisol-producing CYP11B1, which also bind adrenodoxin, revealed substantial structural differences in these regions. The structural and functional differences between different P450 interactions with adrenodoxin may provide valuable clues for an orthogonal treatment approach for primary aldosteronism by specifically targeting the interaction between CYP11B2 and adrenodoxin.
Organizational Affiliation:
Department of Medicinal Chemistry, University of Michigan, Ann Arbor, Michigan, USA.