Structure-Based Drug Discovery ofN-((R)-3-(7-Methyl-1H-indazol-5-yl)-1-oxo-1-(((S)-1-oxo-3-(piperidin-4-yl)-1-(4-(pyridin-4-yl)piperazin-1-yl)propan-2-yl)amino)propan-2-yl)-2'-oxo-1',2'-dihydrospiro[piperidine-4,4'-pyrido[2,3-d][1,3]oxazine]-1-carboxamide (HTL22562): A Calcitonin Gene-Related Peptide Receptor Antagonist for Acute Treatment of Migraine.
Bucknell, S.J., Ator, M.A., Brown, A.J.H., Brown, J., Cansfield, A.D., Cansfield, J.E., Christopher, J.A., Congreve, M., Cseke, G., Deflorian, F., Jones, C.R., Mason, J.S., O'Brien, M.A., Ott, G.R., Pickworth, M., Southall, S.M.(2020) J Med Chem 63: 7906-7920
- PubMed: 32558564 Search on PubMed
- DOI: https://doi.org/10.1021/acs.jmedchem.0c01003
- Primary Citation Related Structures:
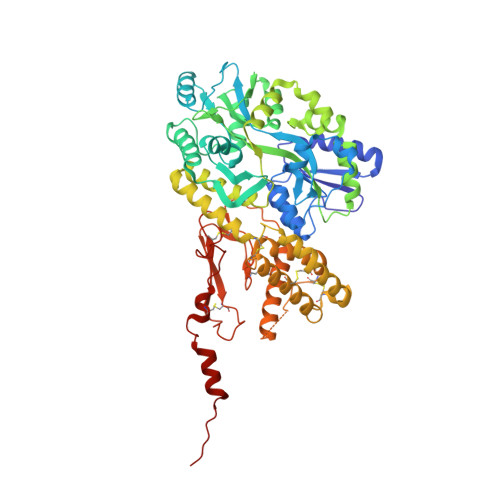
6ZHO, 6ZIS - PubMed Abstract:
Structure-based drug design enabled the discovery of 8 , HTL22562, a calcitonin gene-related peptide (CGRP) receptor antagonist. The structure of 8 complexed with the CGRP receptor was determined at a 1.6 Å resolution. Compound 8 is a highly potent, selective, metabolically stable, and soluble compound suitable for a range of administration routes that have the potential to provide rapid systemic exposures with resultant high levels of receptor coverage (e.g., subcutaneous). The low lipophilicity coupled with a low anticipated clinically efficacious plasma exposure for migraine also suggests a reduced potential for hepatotoxicity. These properties have led to 8 being selected as a clinical candidate for acute treatment of migraine.
- Sosei Heptares, Steinmetz Building, Granta Park, Great Abington, Cambridge CB21 6DG, U.K.
Organizational Affiliation: