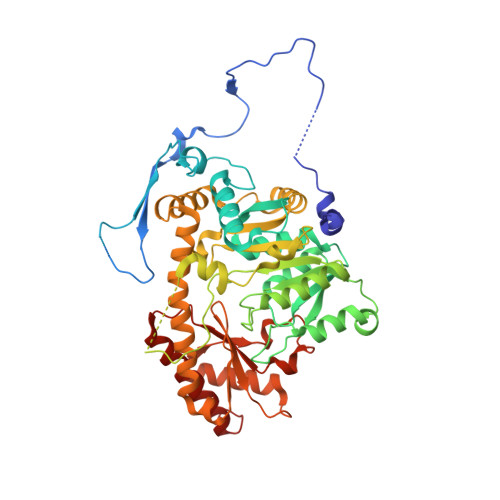
Crystal structures of aminotransferases Aro8 and Aro9 from Candida albicans and structural insights into their properties.
Kiliszek, A., Rypniewski, W., Rzad, K., Milewski, S., Gabriel, I.(2019) J Struct Biol 205: 26-33
- PubMed: 30742897 Search on PubMed
- DOI: https://doi.org/10.1016/j.jsb.2019.02.001
- Primary Citation Related Structures:
6HNB, 6HND, 6HNU, 6HNV - PubMed Abstract:
Aminotransferases catalyze reversibly the transamination reaction by a ping-pong bi-bi mechanism with pyridoxal 5'-phosphate (PLP) as a cofactor. Various aminotransferases acting on a range of substrates have been reported. Aromatic transaminases are able to catalyze the transamination reaction with both aromatic and acidic substrates. Two aminotransferases from C. albicans, Aro8p and Aro9p, have been identified recently, exhibiting different catalytic properties. To elucidate the multiple substrate recognition of the two enzymes we determined the crystal structures of an unliganded CaAro8p, a complex of CaAro8p with the PLP cofactor bound to a substrate, forming an external aldimine, CaAro9p with PLP in the form of internal aldimine, and CaAro9p with a mixture of ligands that have been interpreted as results of the enzymatic reaction. The crystal structures of both enzymes contains in the asymmetric unit a biologically relevant dimer of 55 kDa for CaAro8 and 59 kDa for CaAro9p protein subunits. The ability of the enzymes to process multiple substrates could be related to a feature of their architecture in which the active site resides on one subunit while the substrate-binding site is formed by a long loop extending from the other subunit of the dimeric molecule. The separation of the two functions to different chemical entities could facilitate the evolution of the substrate-binding part and allow it to be flexible without destabilizing the conservative catalytic mechanism.
- Institute of Bioorganic Chemistry, Polish Academy of Sciences, Noskowskiego 12-14, 61-704 Poznań, Poland.
Organizational Affiliation: