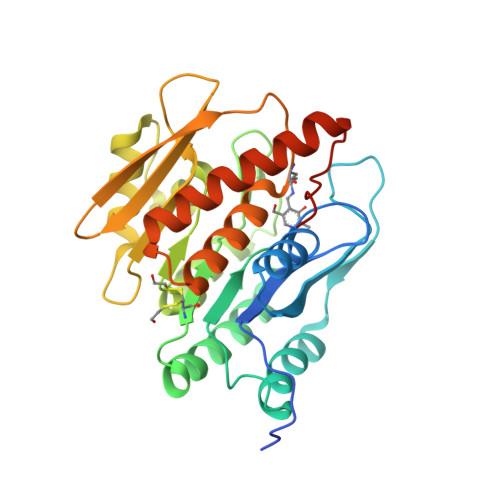
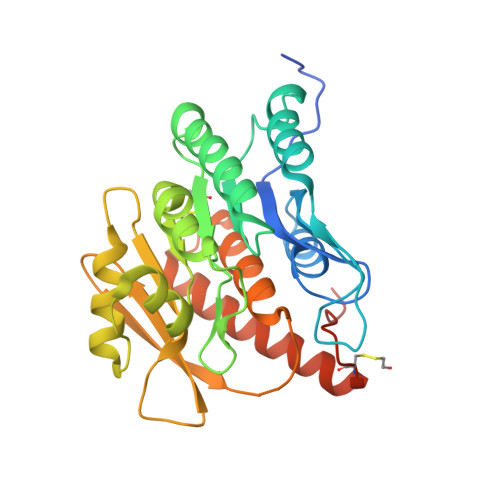
Structural and functional studies on Salmonella typhimurium pyridoxal kinase: the first structural evidence for the formation of Schiff base with the substrate.
Deka, G., Kalyani, J.N., Jahangir, F.B., Sabharwal, P., Savithri, H.S., Murthy, M.R.N.(2019) FEBS J 286: 3684-3700
- PubMed: 31116912 Search on PubMed
- DOI: https://doi.org/10.1111/febs.14933
- Primary Citation Related Structures:
5ZW9, 5ZWA, 5ZWB - PubMed Abstract:
A large number of enzymes depend on the ubiquitous cofactor pyridoxal 5' phosphate (PLP) for their activity. Pyridoxal kinase (PLK) is the key enzyme involved in the synthesis of PLP from the three forms of vitamin B 6 via the salvage pathway. In the present work, we determined the unliganded structure of StPLK in a monoclinic form and its ternary complex with bound pyridoxal (PL), ADP and Mg 2+ in two different tetragonal crystal forms (Form I and Form II). We found that, in the ternary complex structure of StPLK, the active site Lys233 forms a Schiff base linkage with the substrate (PL). Although formation of a Schiff base with the active site Lys229 was demonstrated in the Escherichia coli enzyme based on biochemical studies, the ternary complex of StPLK represents the first crystal structure where the Schiff bond formation has been observed. We also identified an additional site for PLP binding away from the active site in one of the ternary complexes (crystal Form I), suggesting a probable route for the product release. This is the first ternary complex structure where the modeled γ-phosphate of ATP is close enough to PL for the phosphorylation of the substrate. StPLK prefers PL over pyridoxamine as its substrate and follows a sequential mechanism of catalysis. Surface plasmon resonance studies suggest that StPLK interacts with apo-PLP-dependent enzymes with μm affinity supporting the earlier proposed direct transfer mechanism of PLP from PLK to PLP-dependent enzymes.
- Molecular Biophysics Unit, Indian Institute of Science, Bangalore, India.
Organizational Affiliation: