Molecular basis for the specific and multivariant recognitions of RNA substrates by human hnRNP A2/B1
Wu, B.X., Su, S.C., Patil, D.P., Liu, H.H., Gan, J.H., Jaffrey, S.R., Ma, J.B.(2018) Nat Commun
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
(2018) Nat Commun
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Heterogeneous nuclear ribonucleoproteins A2/B1 | 184 | Homo sapiens | Mutation(s): 0 Gene Names: HNRNPA2B1, HNRPA2B1 | 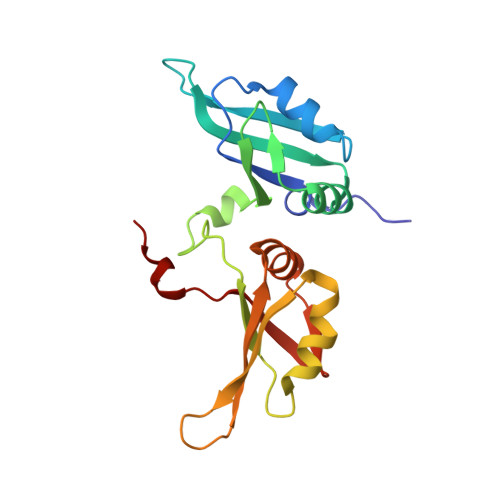 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P22626 GTEx: ENSG00000122566 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P22626 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | ||||
| Molecule | Chains | Length | Organism | Image |
|---|---|---|---|---|
| RNA (5'-R(*AP*GP*GP*AP*CP*UP*G)-3') | 7 | Homo sapiens |  | |
Sequence AnnotationsExpand | ||||
Reference Sequence | ||||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 122.623 | α = 90 |
| b = 46.38 | β = 96.02 |
| c = 37.516 | γ = 90 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| DENZO | data reduction |
| HKL-3000 | data scaling |
| PHASER | phasing |