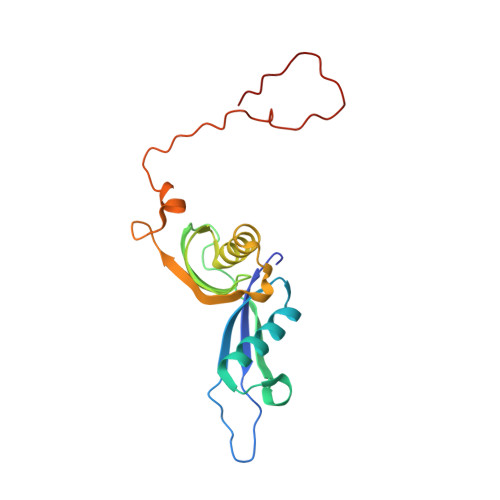
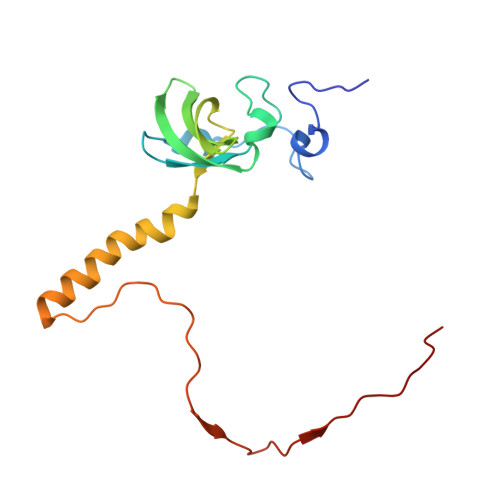
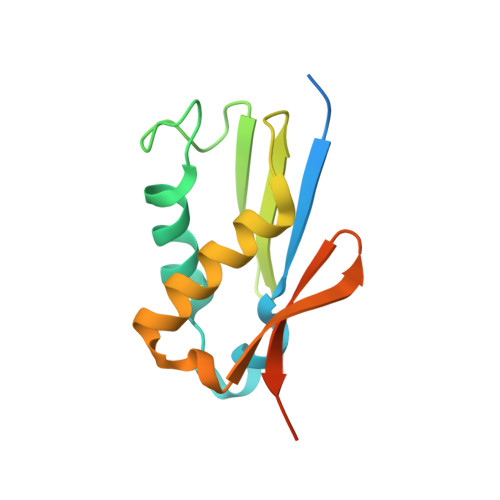
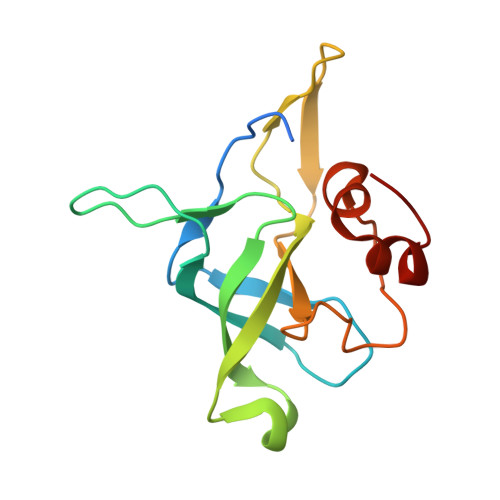
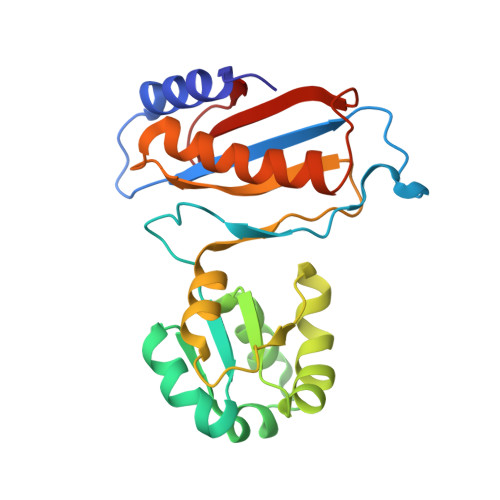
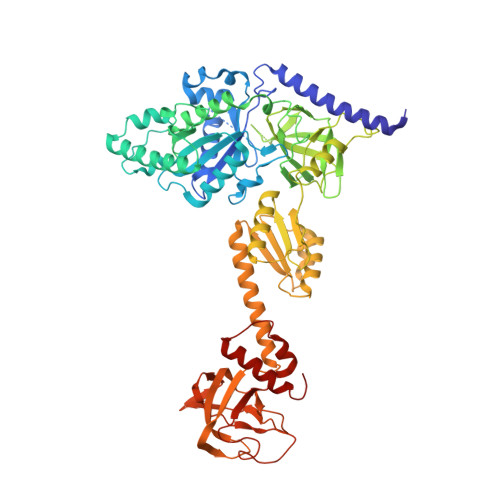
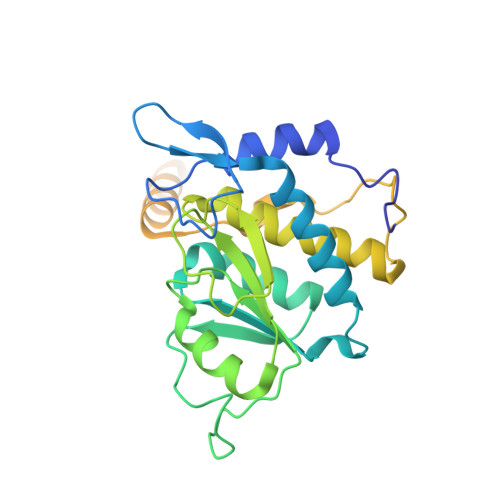
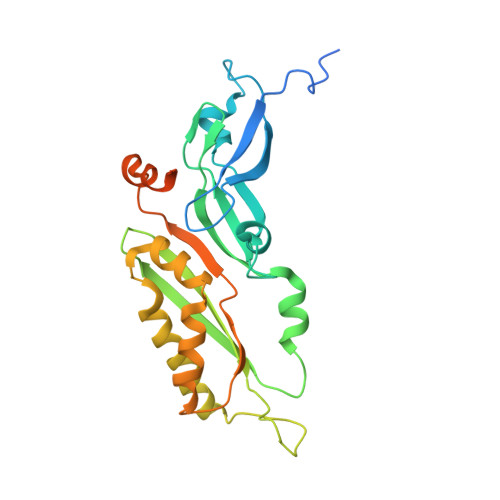
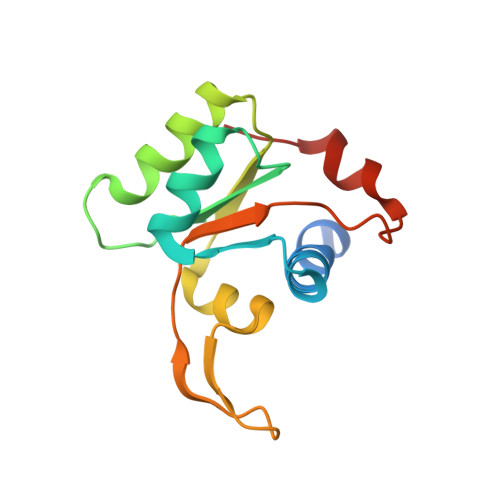
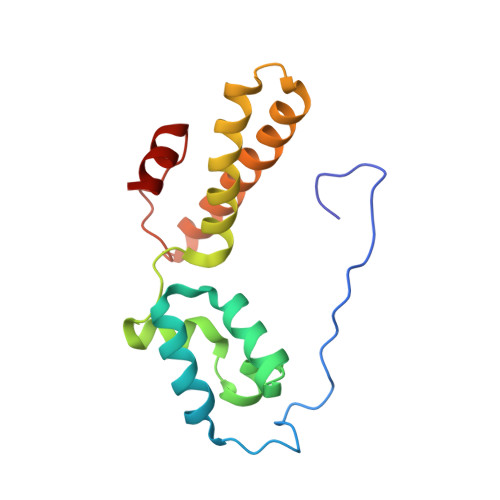
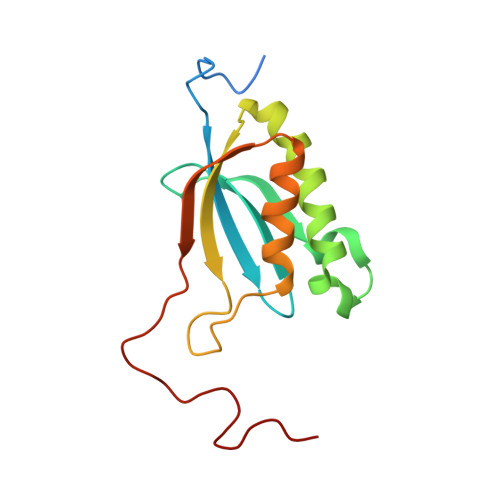
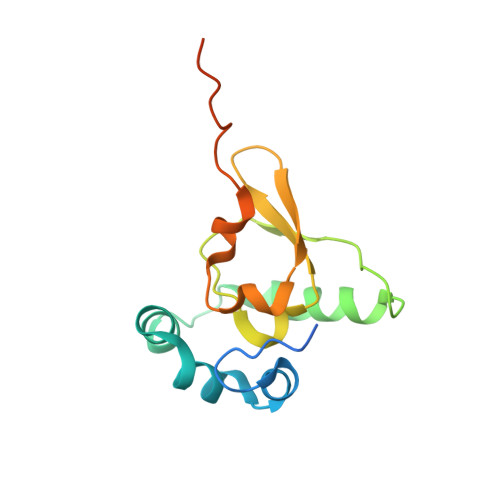
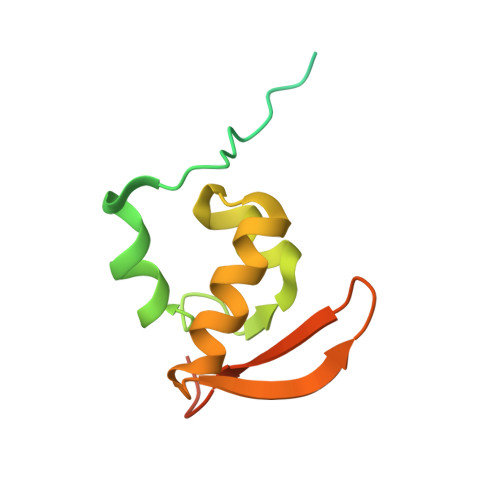
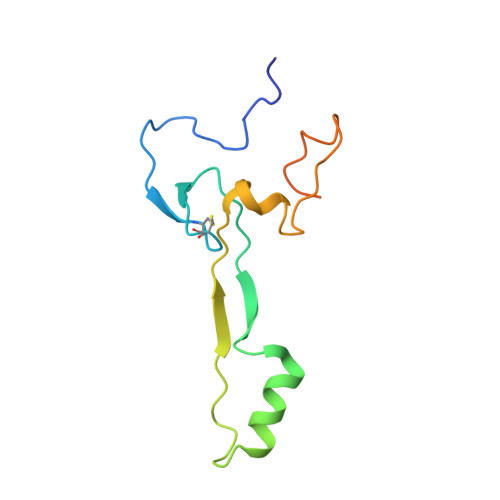
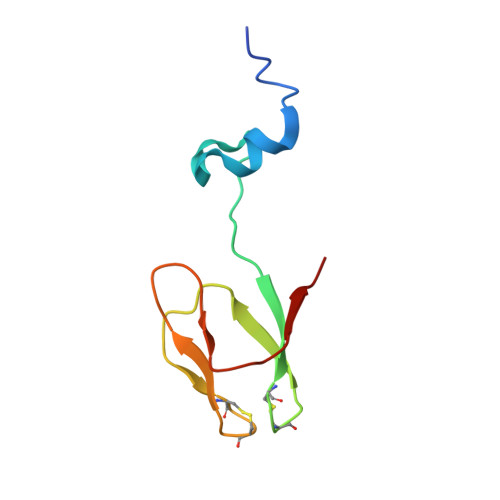
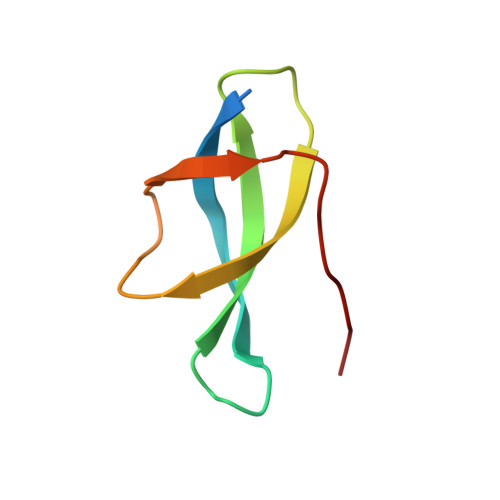
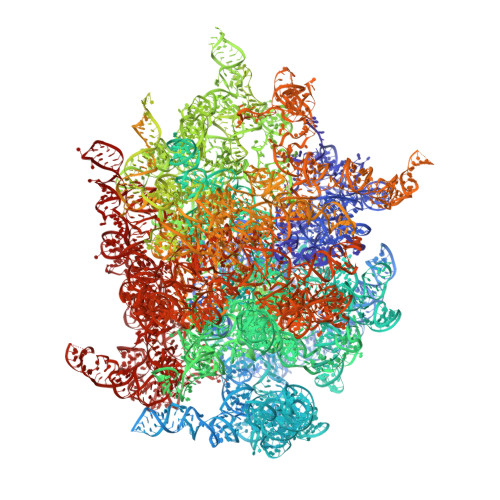
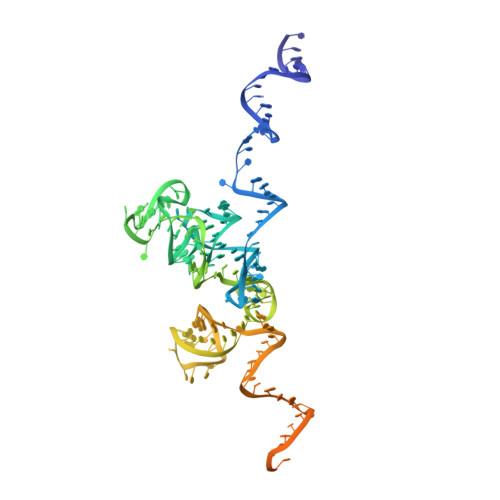
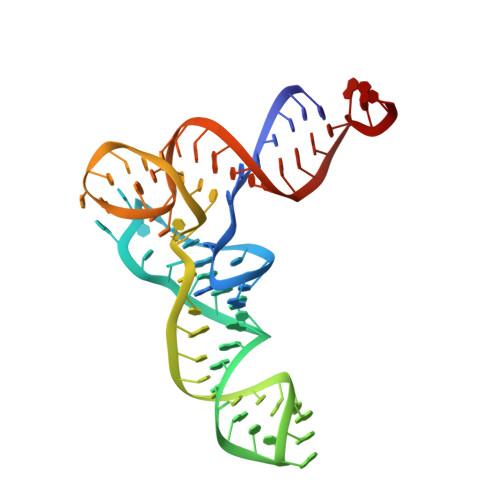
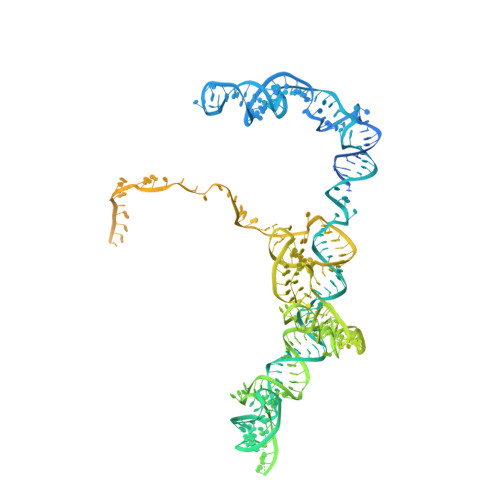
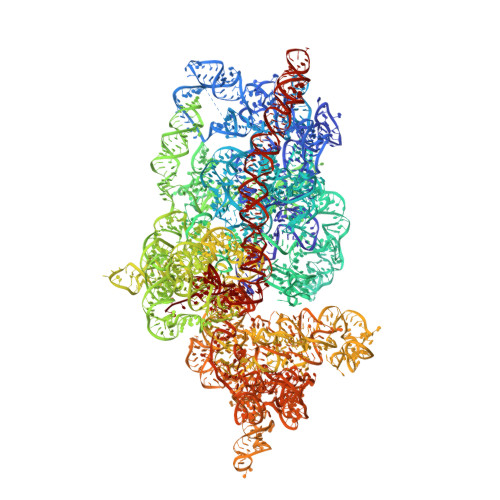
Structure of the Mammalian 80S Initiation Complex with Eif5B on Hcv Ires
Yamamoto, H., Unbehaun, A., Loerke, J., Behrmann, E., Marianne, C., Burger, J., Mielke, T., Spahn, C.M.T.(2014) Nat Struct Mol Biol 21: 721
- PubMed: 25064512 Search on PubMed
- DOI: https://doi.org/10.1038/nsmb.2859
- Primary Citation Related Structures:
4UJC, 4UJD - PubMed Abstract:
The universally conserved eukaryotic initiation factor (eIF) 5B, a translational GTPase, is essential for canonical translation initiation. It is also required for initiation facilitated by the internal ribosomal entry site (IRES) of hepatitis C virus (HCV) RNA. eIF5B promotes joining of 60S ribosomal subunits to 40S ribosomal subunits bound by initiator tRNA (Met-tRNAi(Met)). However, the exact molecular mechanism by which eIF5B acts has not been established. Here we present cryo-EM reconstructions of the mammalian 80S-HCV-IRES-Met-tRNAi(Met)-eIF5B-GMPPNP complex. We obtained two substates distinguished by the rotational state of the ribosomal subunits and the configuration of initiator tRNA in the peptidyl (P) site. Accordingly, a combination of conformational changes in the 80S ribosome and in initiator tRNA facilitates binding of the Met-tRNAi(Met) to the 60S P site and redefines the role of eIF5B as a tRNA-reorientation factor.
- 1] Institute of Medical Physics and Biophysics, Charité-Universitätsmedizin, Berlin, Germany. [2].
Organizational Affiliation: