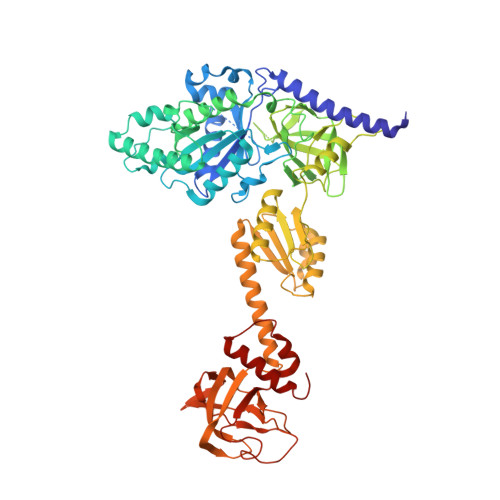
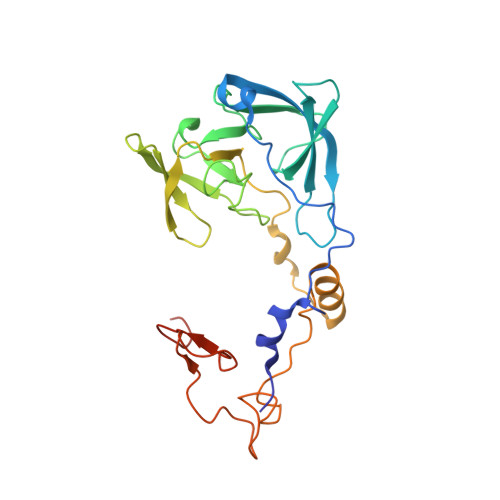
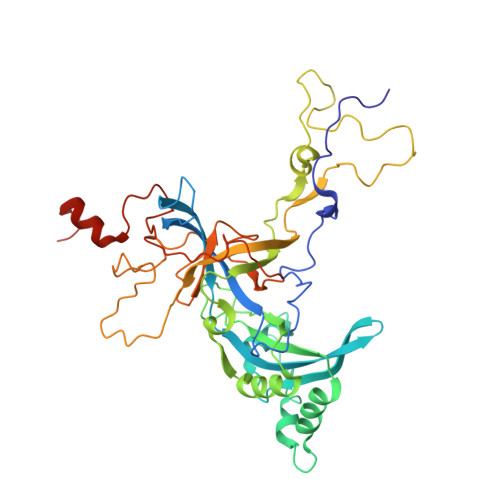
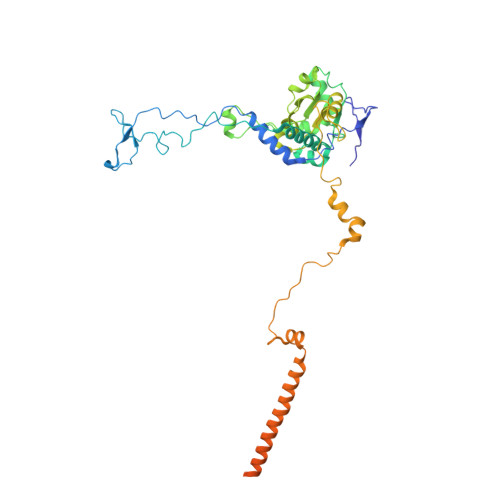
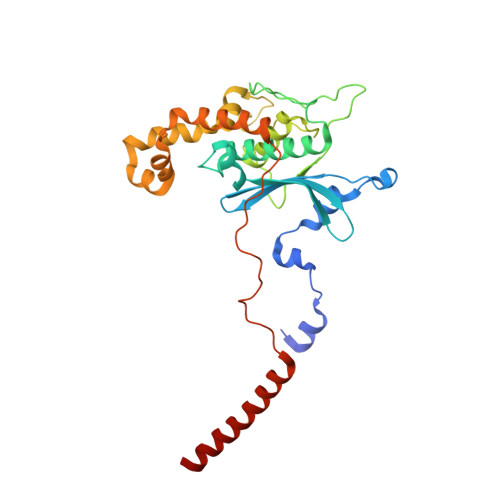
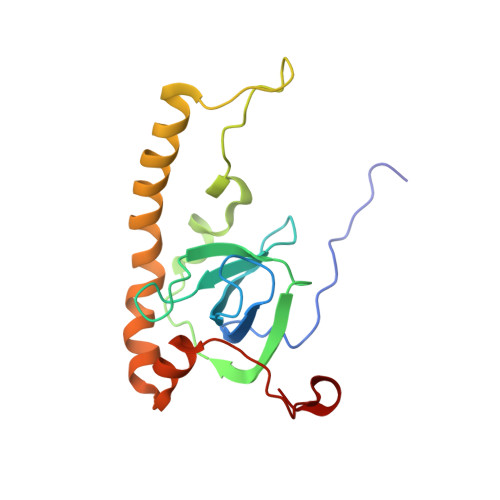
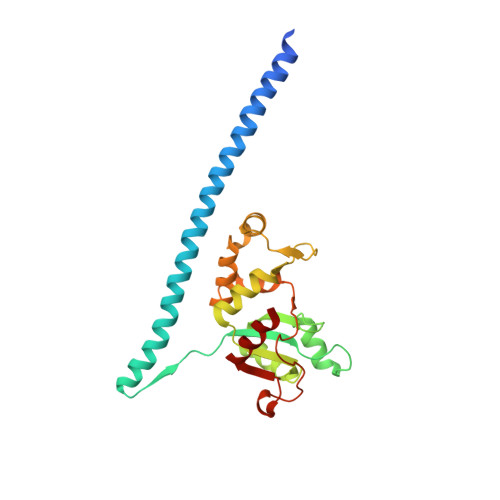
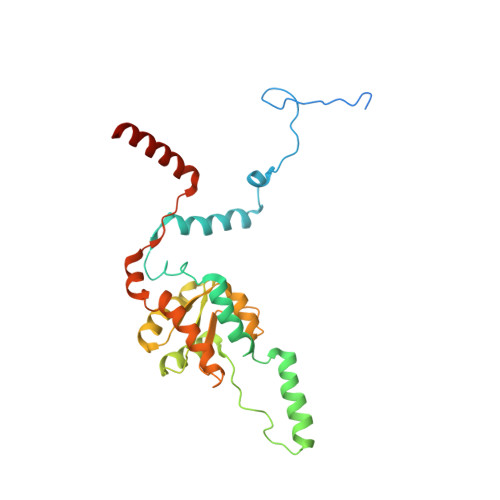
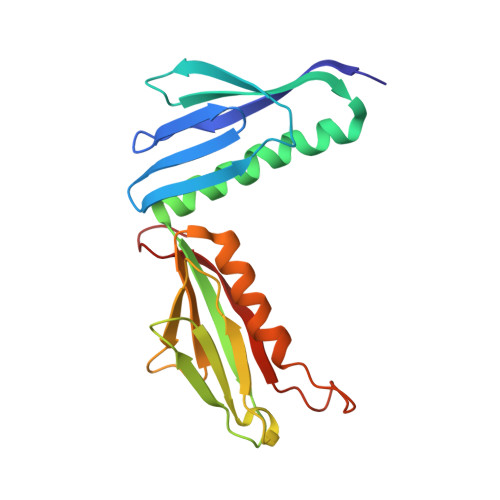
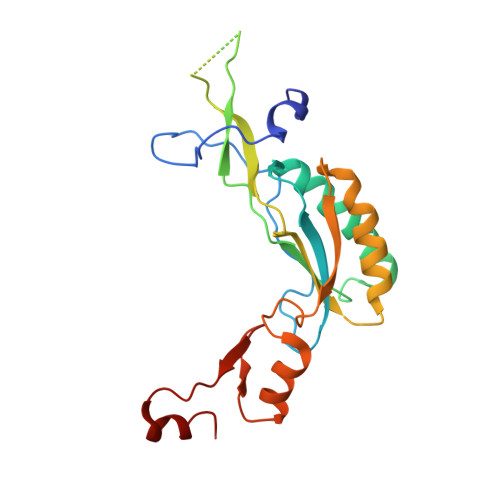
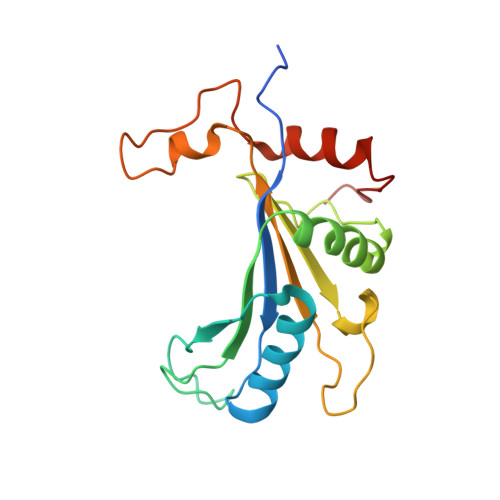
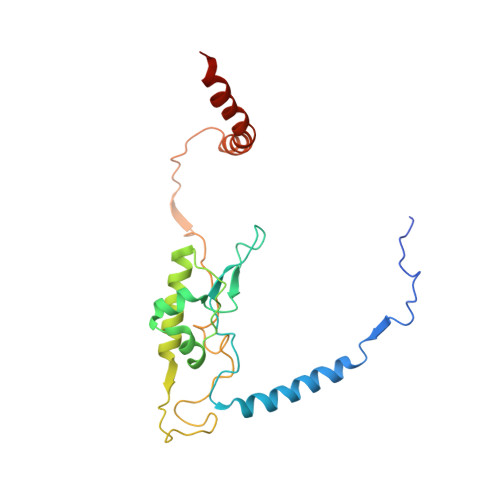
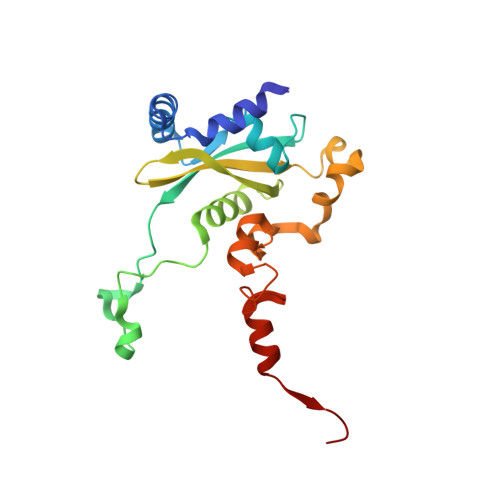
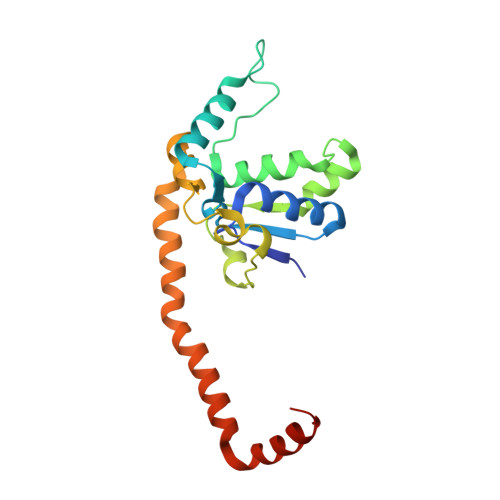
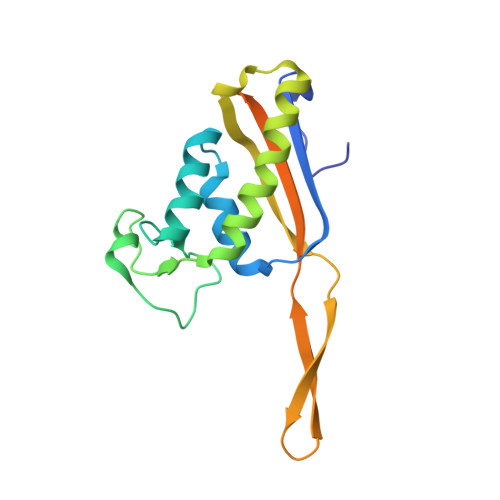
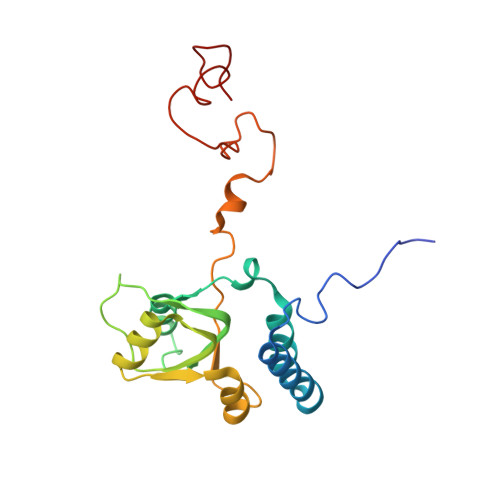
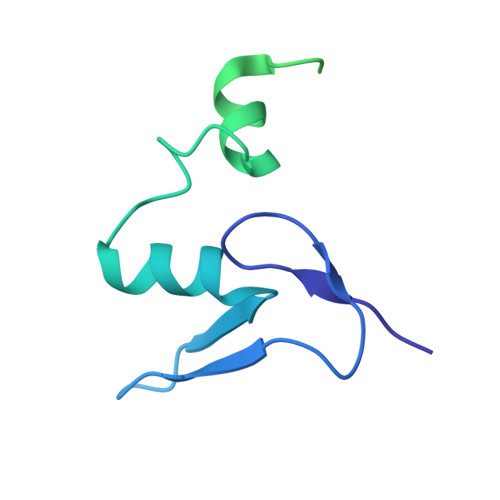
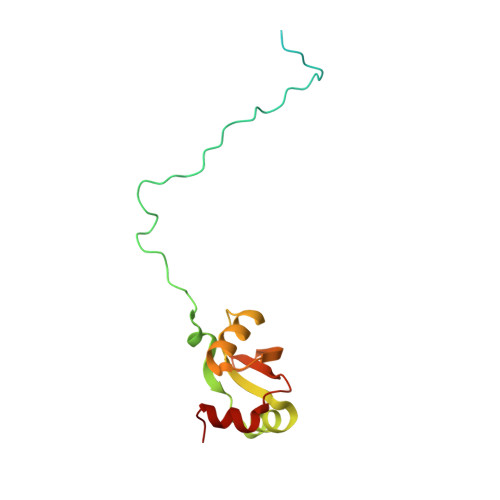
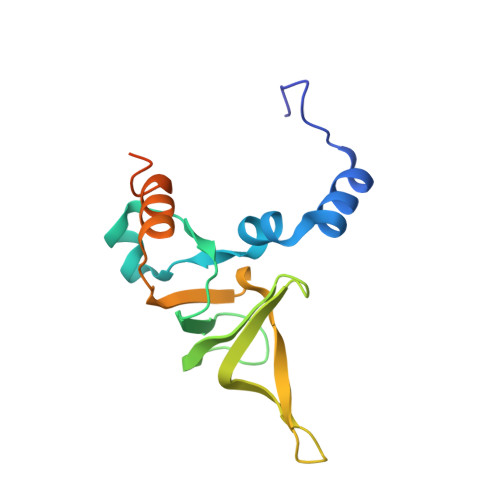
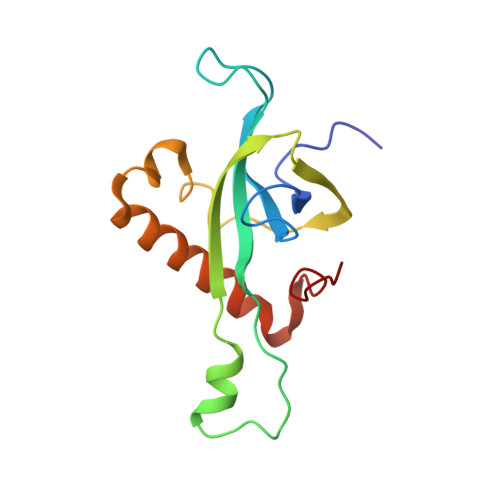
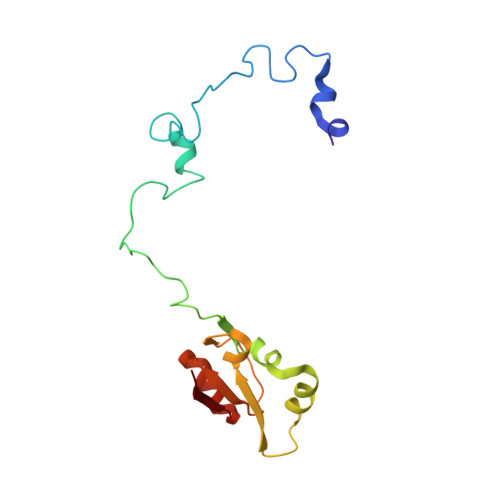
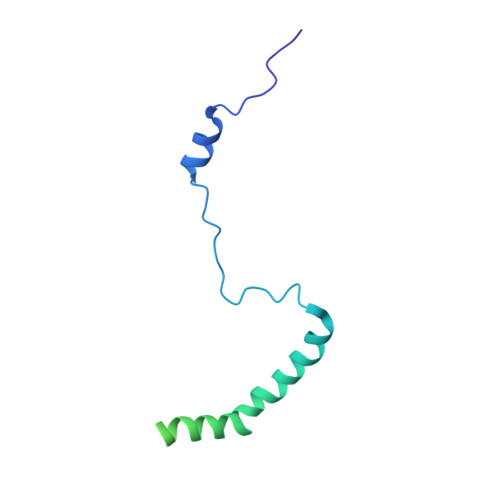
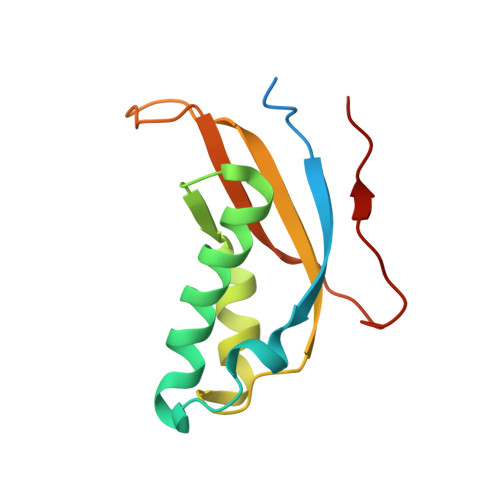
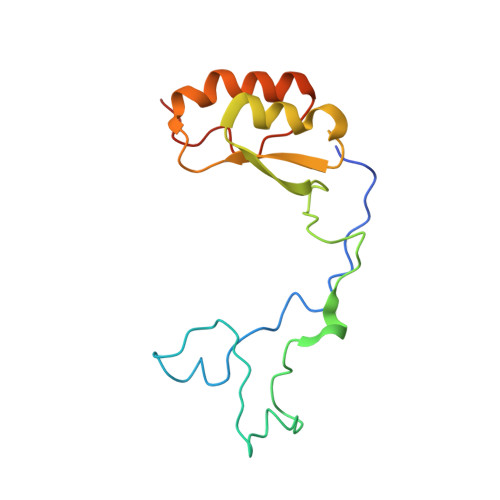
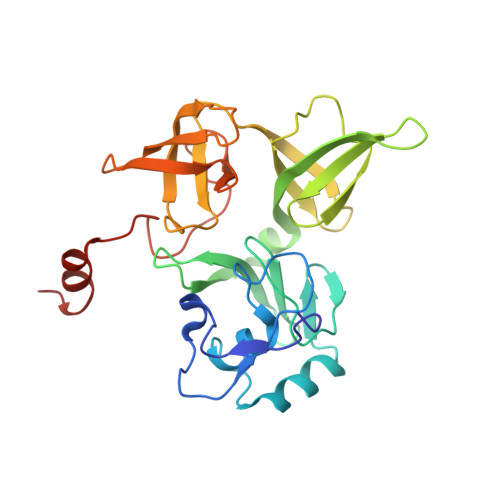
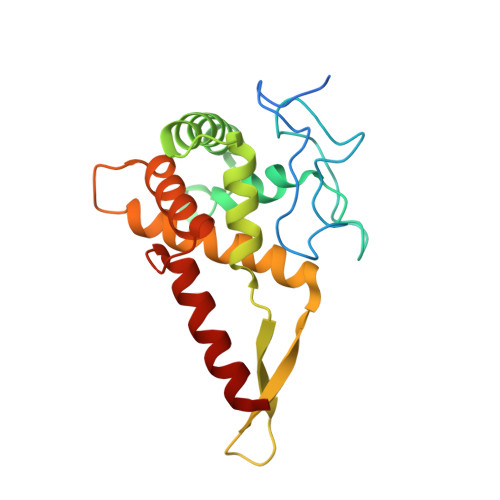
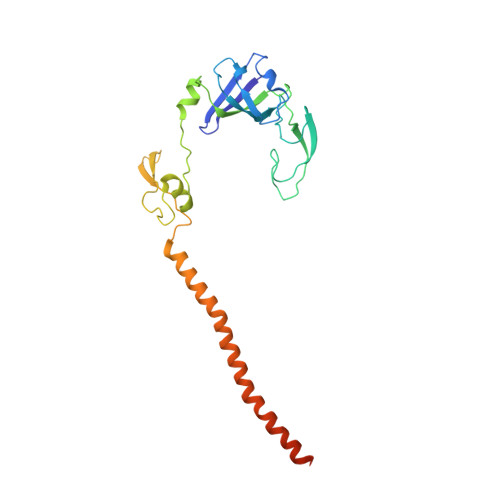
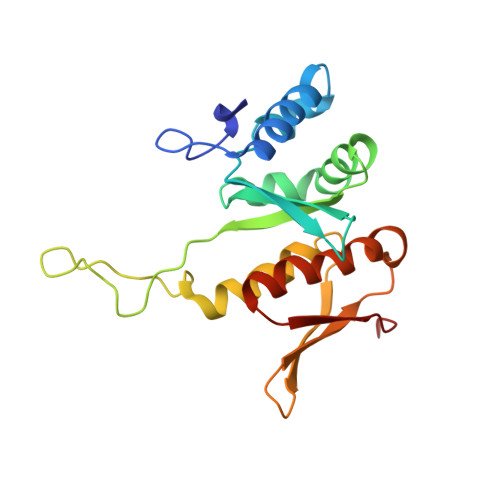
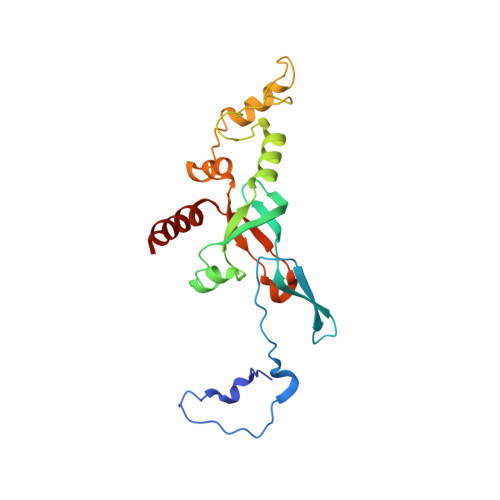
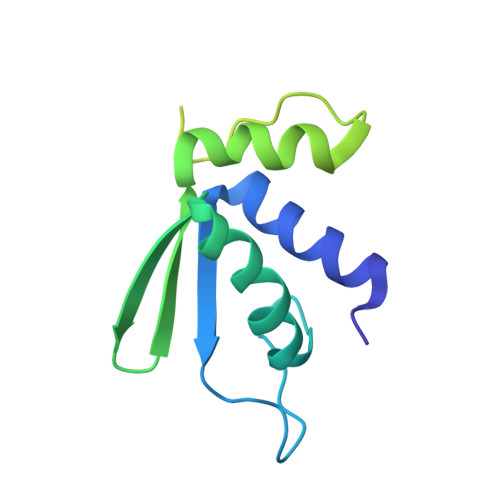
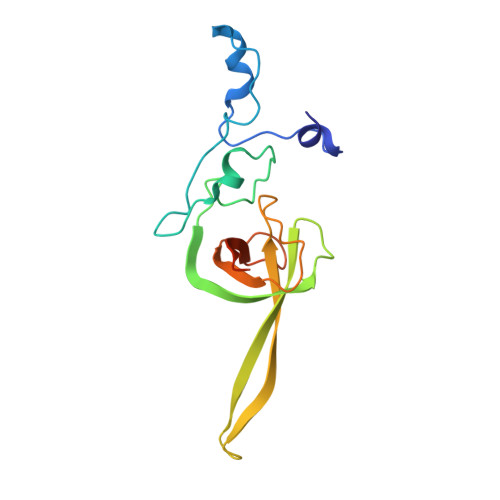
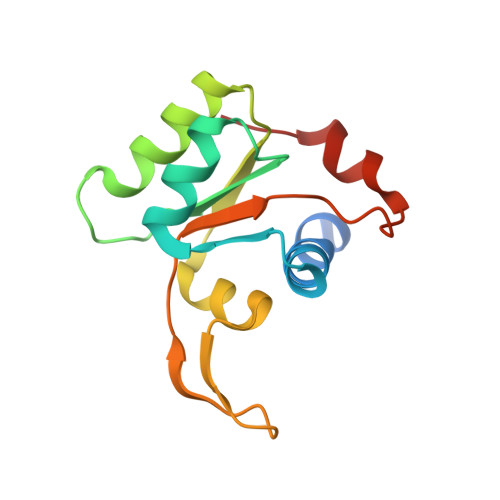
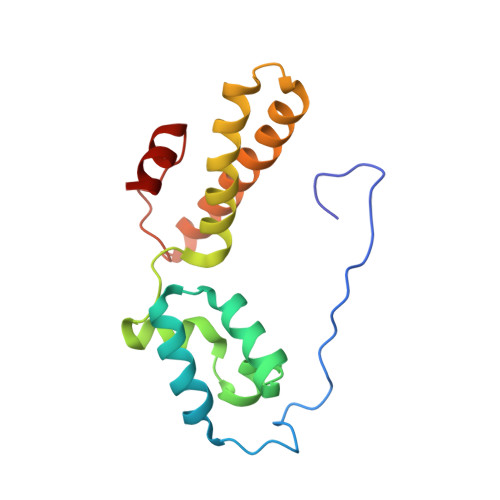
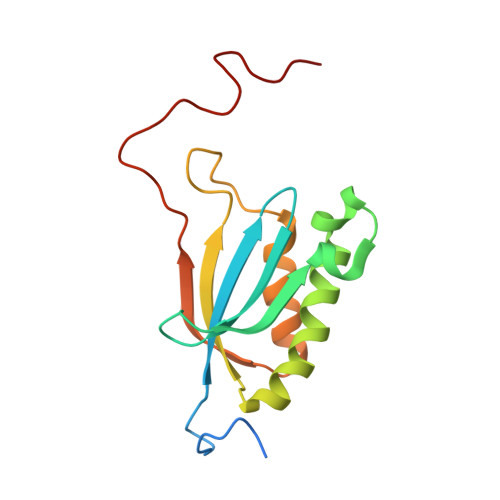
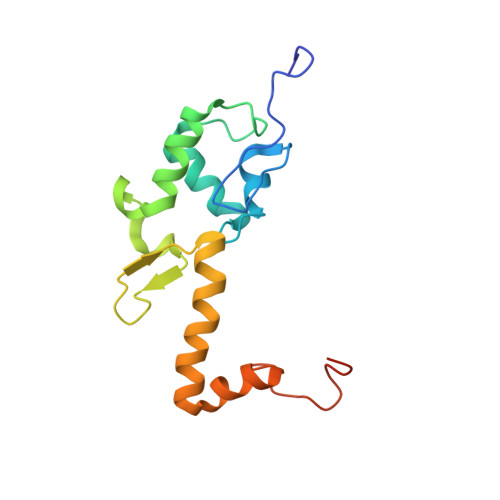
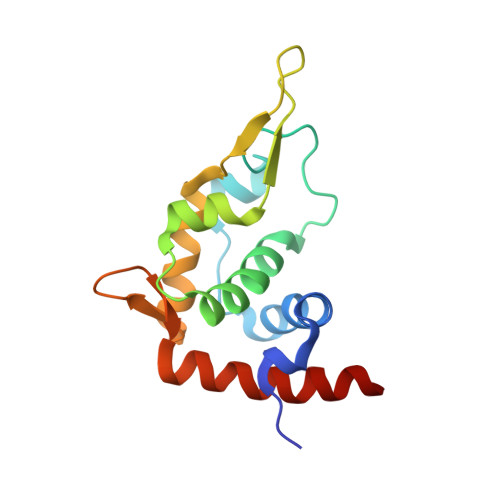
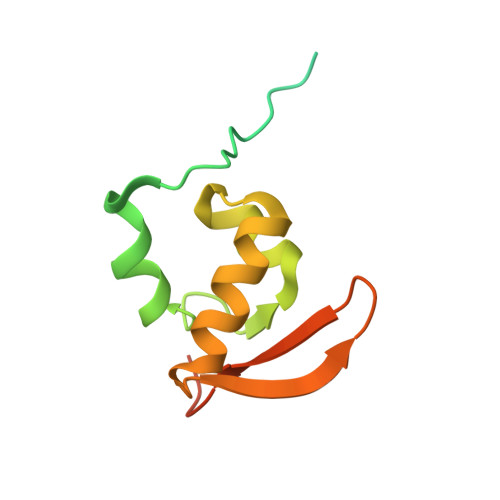
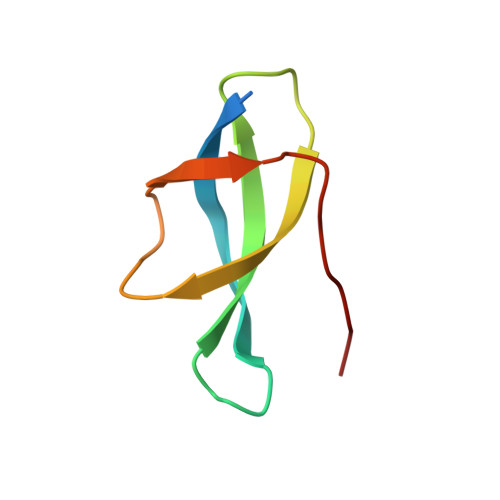
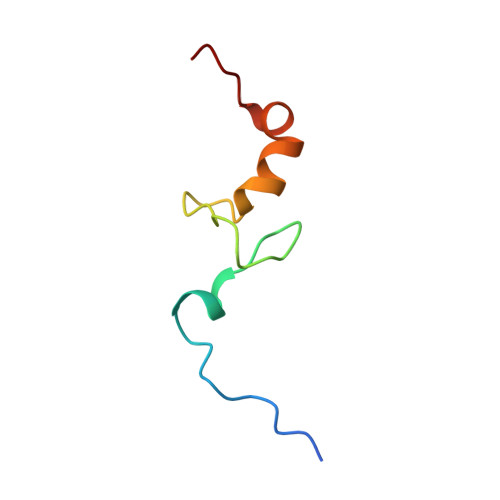
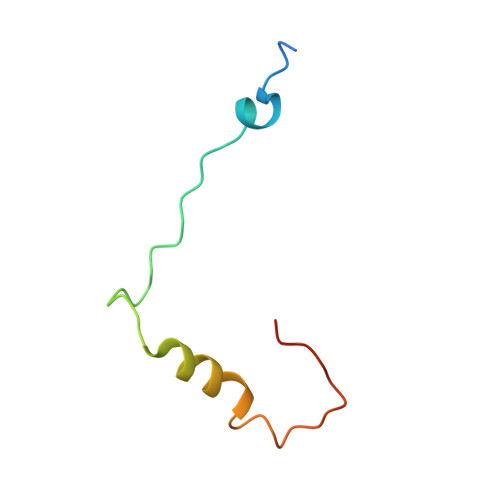
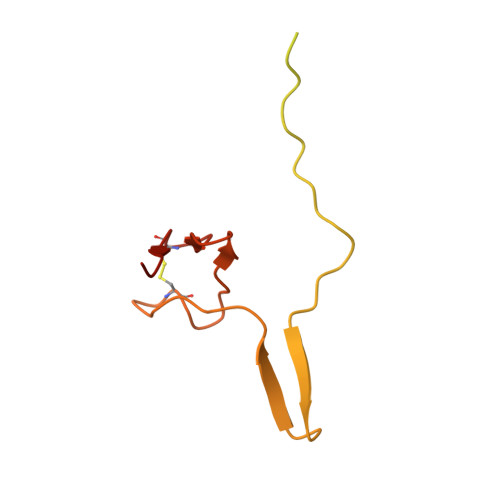
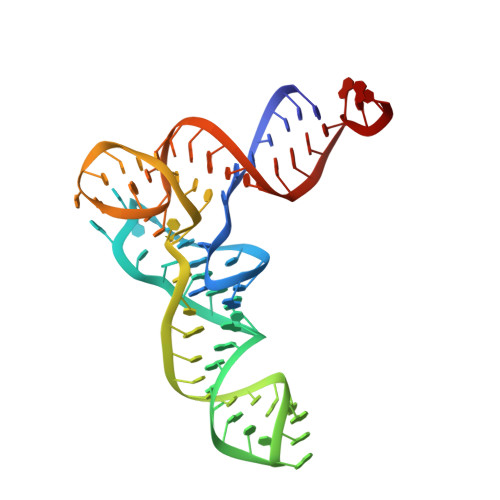
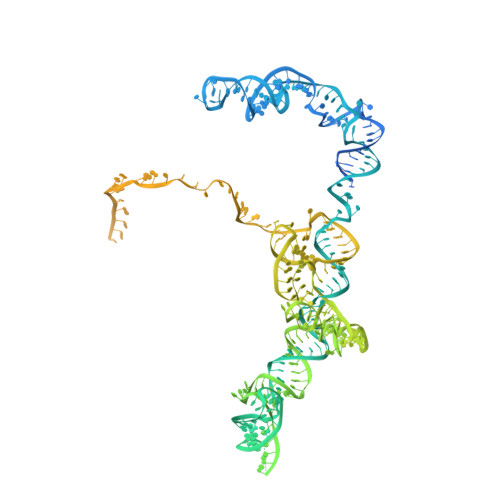
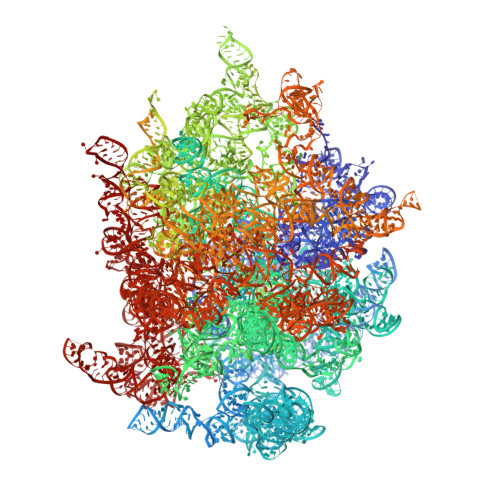
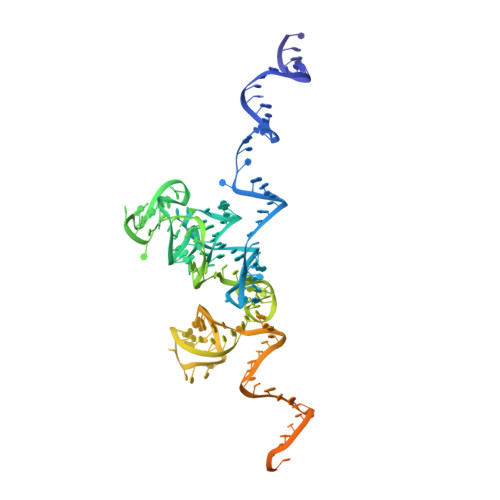
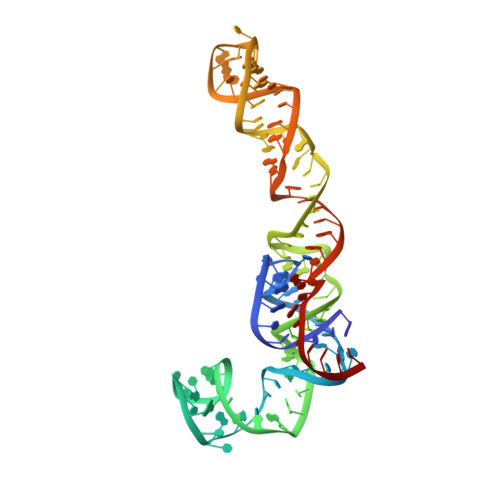
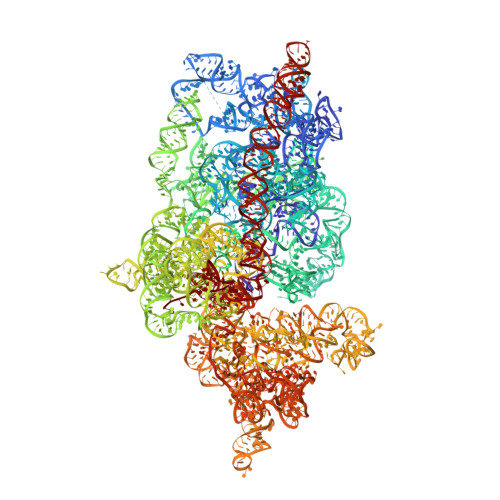
Structure of the Mammalian 80S Initiation Complex with Initiation Factor 5B on Hcv-Ires RNA.
Yamamoto, H., Unbehaun, A., Loerke, J., Behrmann, E., Collier, M., Burger, J., Mielke, T., Spahn, C.M.T.(2014) Nat Struct Mol Biol 21: 721
- PubMed: 25064512 Search on PubMed
- DOI: https://doi.org/10.1038/nsmb.2859
- Primary Citation Related Structures:
4UJC, 4UJD - PubMed Abstract:
The universally conserved eukaryotic initiation factor (eIF) 5B, a translational GTPase, is essential for canonical translation initiation. It is also required for initiation facilitated by the internal ribosomal entry site (IRES) of hepatitis C virus (HCV) RNA. eIF5B promotes joining of 60S ribosomal subunits to 40S ribosomal subunits bound by initiator tRNA (Met-tRNAi(Met)). However, the exact molecular mechanism by which eIF5B acts has not been established. Here we present cryo-EM reconstructions of the mammalian 80S-HCV-IRES-Met-tRNAi(Met)-eIF5B-GMPPNP complex. We obtained two substates distinguished by the rotational state of the ribosomal subunits and the configuration of initiator tRNA in the peptidyl (P) site. Accordingly, a combination of conformational changes in the 80S ribosome and in initiator tRNA facilitates binding of the Met-tRNAi(Met) to the 60S P site and redefines the role of eIF5B as a tRNA-reorientation factor.
- 1] Institute of Medical Physics and Biophysics, Charité-Universitätsmedizin, Berlin, Germany. [2].
Organizational Affiliation: