Crystal structure of human SET domain-containing protein3
Zeng, H., Dong, A., Walker, J.R., Loppnau, P., Bountra, C., Weigelt, J., Arrowsmith, C.H., Edwards, A.M., Min, J., Wu, H.To be published.
Experimental Data Snapshot
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Histone-lysine N-methyltransferase setd3 | 497 | Homo sapiens | Mutation(s): 0 Gene Names: SETD3, C14orf154 EC: 2.1.1.43 (PDB Primary Data), 2.1.1.85 (UniProt) | 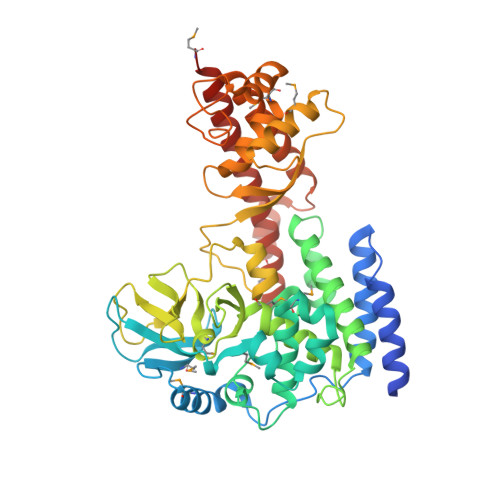 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q86TU7 GTEx: ENSG00000183576 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q86TU7 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 5 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| SAM Download:Ideal Coordinates CCD File | B [auth A] | S-ADENOSYLMETHIONINE C15 H22 N6 O5 S MEFKEPWMEQBLKI-FCKMPRQPSA-N |  | ||
| PG4 Download:Ideal Coordinates CCD File | E [auth A] | TETRAETHYLENE GLYCOL C8 H18 O5 UWHCKJMYHZGTIT-UHFFFAOYSA-N |  | ||
| ARS Download:Ideal Coordinates CCD File | C [auth A], D [auth A] | ARSENIC As RBFQJDQYXXHULB-UHFFFAOYSA-N |  | ||
| ACT Download:Ideal Coordinates CCD File | F [auth A], G [auth A] | ACETATE ION C2 H3 O2 QTBSBXVTEAMEQO-UHFFFAOYSA-M |  | ||
| UNX Download:Ideal Coordinates CCD File | H [auth A] I [auth A] J [auth A] K [auth A] L [auth A] | UNKNOWN ATOM OR ION X |  | ||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| MSE Query on MSE | A | L-PEPTIDE LINKING | C5 H11 N O2 Se |  | MET |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 113.864 | α = 90 |
| b = 113.864 | β = 90 |
| c = 83.57 | γ = 120 |
| Software Name | Purpose |
|---|---|
| SBC-Collect | data collection |
| MOLREP | phasing |
| BUSTER | refinement |
| Coot | model building |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |