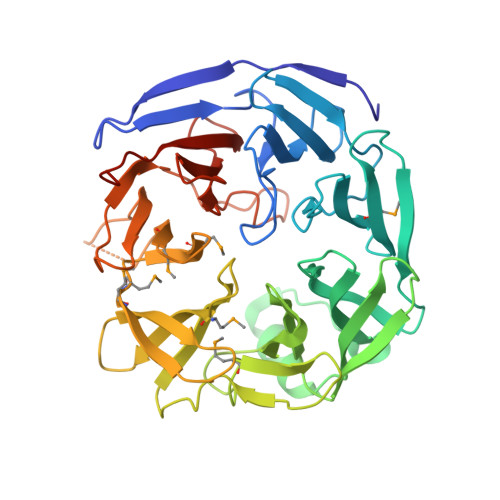
Crystal structure of oligogalacturonate lyase (VPA0088) from Vibrio parahaemolyticus.
Forouhar, F., Abashidze, M., Seetharaman, J., Janjua, H., Mao, L., Xiao, R., Owens, L.A., Wang, D., Baran, M.C., Acton, T.B., Montelione, G.T., Hunt, J.F., Tong, L.To be published.