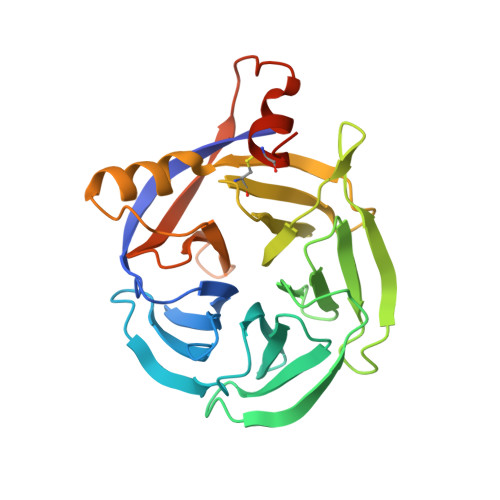
Crystal structure of papaya glutaminyl cyclase, an archetype for plant and bacterial glutaminyl cyclases.
Wintjens, R., Belrhali, H., Clantin, B., Azarkan, M., Bompard, C., Baeyens-Volant, D., Looze, Y., Villeret, V.(2006) J Mol Biology 357: 457-470
- PubMed: 16438985 Search on PubMed
- DOI: https://doi.org/10.1016/j.jmb.2005.12.029
- Primary Citation Related Structures:
2FAW - PubMed Abstract:
Glutaminyl cyclases (QCs) (EC 2.3.2.5) catalyze the intramolecular cyclization of protein N-terminal glutamine residues into pyroglutamic acid with the concomitant liberation of ammonia. QCs may be classified in two groups containing, respectively, the mammalian enzymes, and the enzymes from plants, bacteria, and parasites. The crystal structure of the QC from the latex of Carica papaya (PQC) has been determined at 1.7A resolution. The structure was solved by the single wavelength anomalous diffraction technique using sulfur and zinc as anomalous scatterers. The enzyme folds into a five-bladed beta-propeller, with two additional alpha-helices and one beta hairpin. The propeller closure is achieved via an original molecular velcro, which links the last two blades into a large eight stranded beta-sheet. The zinc ion present in the PQC is bound via an octahedral coordination into an elongated cavity located along the pseudo 5-fold axis of the beta-propeller fold. This zinc ion presumably plays a structural role and may contribute to the exceptional stability of PQC, along with an extended hydrophobic packing, the absence of long loops, the three-joint molecular velcro and the overall folding itself. Multiple sequence alignments combined with structural analyses have allowed us to tentatively locate the active site, which is filled in the crystal structure either by a Tris molecule or an acetate ion. These analyses are further supported by the experimental evidence that Tris is a competitive inhibitor of PQC. The active site is located at the C-terminal entrance of the PQC central tunnel. W83, W110, W169, Q24, E69, N155, K225, F22 and F67 are highly conserved residues in the C-terminal entrance, and their putative role in catalysis is discussed. The PQC structure is representative of the plants, bacterial and parasite enzymes and contrasts with that of mammalian enzymes, that may possibly share a conserved scaffold of the bacterial aminopeptidase.
- Laboratoire de Chimie Générale, Institut de Pharmacie-U.L.B. CP 206/04, Boulevard du Triomphe, B-1050 Brussels, Belgium.
Organizational Affiliation: