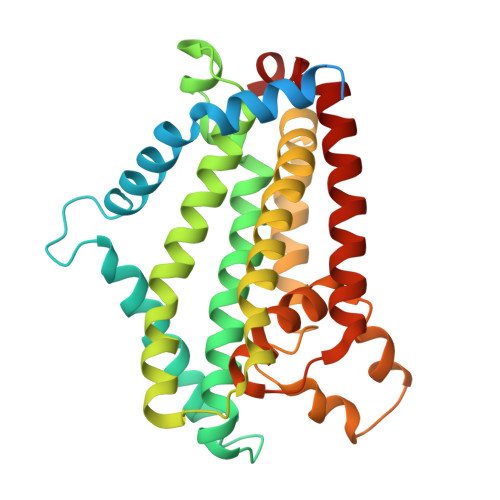
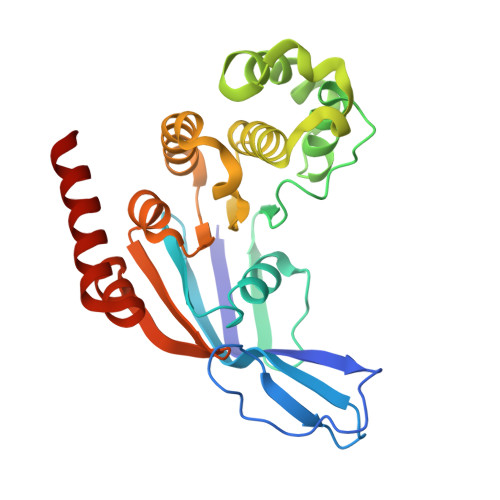
Structural basis of lipid-linked galactan export by the mycobacterial ABC transporter Wzm-Wzt.
Garaeva, A.A., Fabianova, V., Savkova, K., Huszar, S., Xue, X., Lowary, T.L., Mikusova, K., Seeger, M.A.(2026) Nat Commun 17
- PubMed: 41839883 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-026-70429-9
- Primary Citation Related Structures:
9QFX, 9QGU, 9QH1, 9QHJ, 9QHK, 9QHV, 9QHW, 9QHX - PubMed Abstract:
Mycobacteria, including Mycobacterium tuberculosis, possess a unique cell envelope containing arabinogalactan, a heteropolysaccharide critical for cell wall integrity and target of several tuberculosis drugs. The cytosolic precursor of arabinogalactan, lipid-linked galactan (LLG), is translocated across the plasma membrane by the essential ABC transporter Wzm-Wzt through a molecular mechanism that is poorly understood. Here, we present a series of cryo-EM structures of Wzm-Wzt from Mycobacterium abscessus, representing different conformations of the transport cycle. Conserved residues lining the proposed LLG translocation pathway were investigated by three orthologous functional assays, revealing that the cytosolic gate helix (GH) plays a key functional role in polysaccharide transport. Our data suggests that the hydrophobic polyprenyl-moiety is translocated first, followed by the galactan-polysaccharide, which requires Wzm-Wzt to open a continuous channel through which the sugar chain is ratcheted at the expense of ATP hydrolysis. Our results provide a rational basis for the development of drugs that inhibit mycobacterial cell wall biosynthesis.
- Institute of Medical Microbiology, University of Zurich, Zurich, Switzerland. alisa.garaeva@uzh.ch.
Organizational Affiliation: