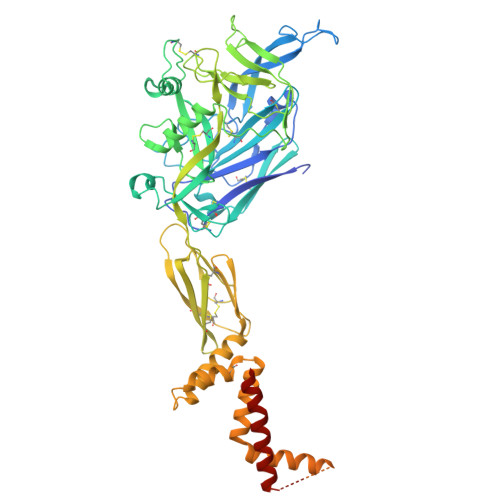
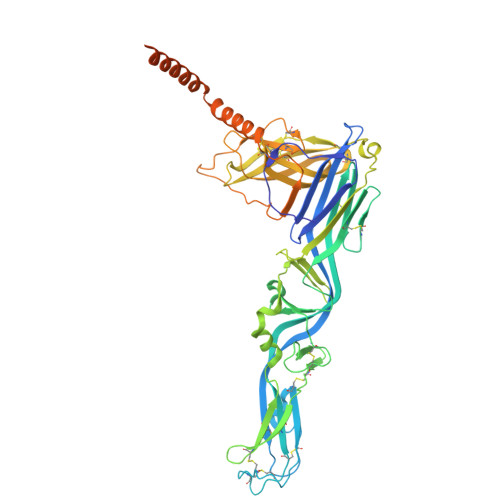
High-resolution in situ structures of hantavirus glycoprotein tetramers.
Guo, L., McFadden, E., Slough, M.M., Stone, E.T., Berrigan, J., Mittler, E., Hatzakis, K., Hinkley, T., Kain, H.S., Ke, Z., Warner, N.L., Erasmus, J.H., Chandran, K., McLellan, J.S.(2026) Cell 189: 2731
- PubMed: 41763200 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.cell.2026.01.030
- Primary Citation Related Structures:
9P3I, 9P3L, 9P3M, 9P3X, 9P3Y - PubMed Abstract:
New World hantaviruses cause severe infections in humans. Previous structural studies have advanced our understanding of hantavirus glycoprotein architecture; however, the lack of high-resolution structures of the glycoprotein tetramer and its lattice organization has limited mechanistic insights into viral assembly and entry. Here, we leveraged a virus-like particle (VLP) system to establish a cryo-electron microscopy workflow for lattice-forming viral glycoproteins. This enabled the determination of a 2.35 Å resolution structure of the membrane-embedded Andes virus (ANDV) glycoprotein tetramer as well as the structures of dimers of tetramers and a complex with antibody ADI-65534. These structures reveal previously uncharacterized features of glycoprotein organization, stability, and pH sensing. The immunization of mice with self-amplifying replicon RNA (repRNA) encoding ANDV-VLPs elicited high levels of glycoprotein-binding antibodies but equivalent titers of neutralizing antibodies compared with the repRNA-encoded native ANDV glycoprotein complex. These findings advance our understanding of hantavirus glycoprotein assemblies, laying a foundation for structure-based vaccine design.
- Department of Molecular Biosciences, The University of Texas at Austin, Austin, TX 78712, USA.
Organizational Affiliation: