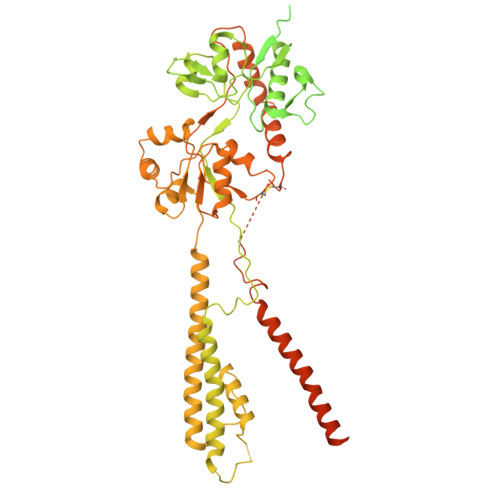
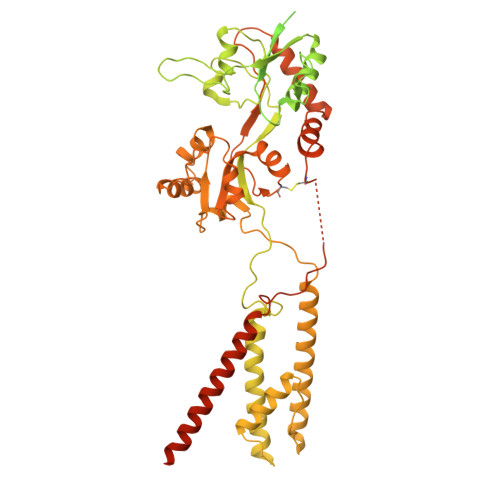
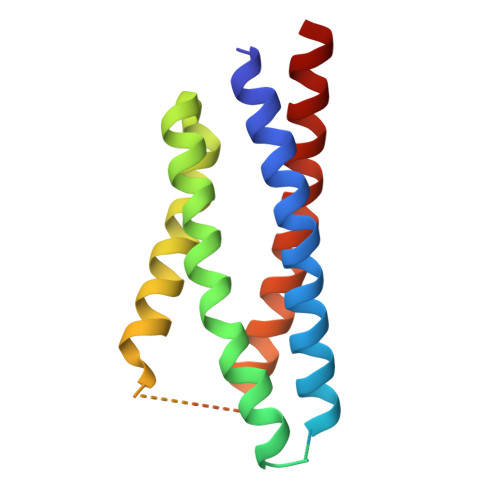
Assembly and gating mechanism of native AMPA receptors from the cerebellum.
Li, X., Li, R., Wei, Y., Chen, J., Zhao, J., Zhao, J., Wang, W., Li, N., Wang, L., Hu, T., Dong, Y., Zhu, Y., Wei, C., Li, L., Zhang, W., Huang, Z., Zhao, Y.(2026) Cell Res
- PubMed: 41840198 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41422-026-01234-8
- Primary Citation Related Structures:
9J92, 9J93, 9J94, 9J95, 9X57, 9X58, 9XJL, 9XJM - PubMed Abstract:
AMPA receptors (AMPARs) mediate the majority of fast excitatory synaptic transmission throughout the central nervous system. Calcium-permeable AMPARs and GluA4-containing receptors are critical for cerebellar functions, such as motor learning, associative memory, auditory processing, and synaptic plasticity. In contrast to the well-characterized, predominantly GluA2-containing AMPARs of the hippocampus and cortex, cerebellar AMPARs contain a higher proportion of GluA4 and remain poorly understood. Here, we generated a highly GluA4-specific antibody. Using this antibody in combination with antibodies specifically recognizing GluA1 and GluA2, we purified native AMPARs and determined the subunit compositions of both calcium-impermeable and calcium-permeable native AMPARs in the cerebellum. The isolated cerebellar AMPARs that contained both GluA1 and GluA4 were calcium-permeable, with GluA4 occupying mainly the B/D positions, GluA1 occupying the A/C positions, and the complex associated primarily with cornichon 3 (CNIH3). We determined the structures of the complex in distinct functional states, including the resting, active, and desensitized states, and characterized the conformational transitions that underlie its activity. During desensitization, the receptor adopts a pseudo-4-fold configuration of the ligand-binding domain layer, which may be important for its functional properties. This study provides a blueprint for the subunit compositions of AMPARs in the cerebellum and clarifies the gating mechanism of the calcium-permeable native AMPAR A1A4 -CNIH3 complex, providing significant insight into AMPAR-mediated synaptic transmission in the cerebellum.
- Key Laboratory of Biomacromolecules (CAS), National Laboratory of Biomacromolecules, CAS Center for Excellence in Biomacromolecules, Institute of Biophysics, Chinese Academy of Sciences, Beijing, China.
Organizational Affiliation: