Crystal structure of C35 bound to Hem
Marchesani, F., De Bei, O., Brear, P., Spyrakis, F., Lazzarato, L., Ronda, L.To be published.
Experimental Data Snapshot
Starting Model: in silico
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Hemoglobin subunit alpha | 141 | Homo sapiens | Mutation(s): 0 Gene Names: HBA1, HBA2 | 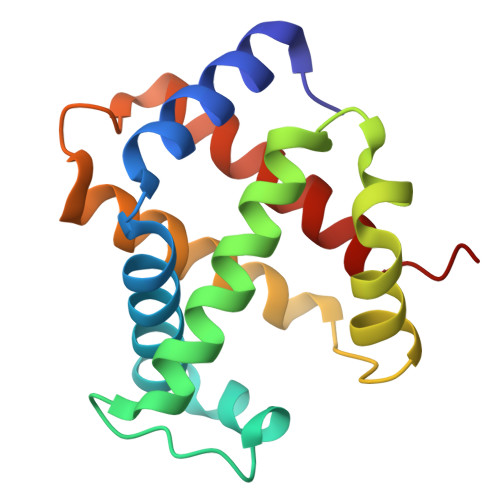 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P69905 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P69905 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Hemoglobin subunit beta | 146 | Homo sapiens | Mutation(s): 0 Gene Names: HBB | 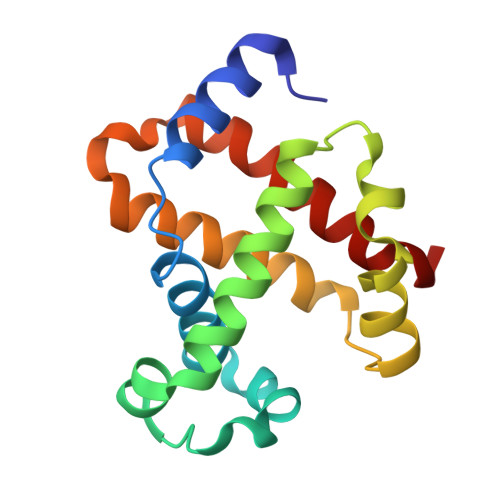 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P68871 GTEx: ENSG00000244734 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P68871 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 8 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| HEM Download:Ideal Coordinates CCD File | E [auth A], JA [auth C], QA [auth D], S [auth B] | PROTOPORPHYRIN IX CONTAINING FE C34 H32 Fe N4 O4 KABFMIBPWCXCRK-RGGAHWMASA-L |  | ||
| A1ITR (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | G [auth A], HA [auth C], U [auth B] | 4-[2-[[5-(1H-indol-3-yl)-1,3,4-oxadiazol-2-yl]sulfanyl]ethanoylamino]benzoic acid C19 H14 N4 O4 S DHEYLYPADJBRNO-UHFFFAOYSA-N |  | ||
| PGE Download:Ideal Coordinates CCD File | X [auth B] | TRIETHYLENE GLYCOL C6 H14 O4 ZIBGPFATKBEMQZ-UHFFFAOYSA-N |  | ||
| SO4 Download:Ideal Coordinates CCD File | AA [auth B] BA [auth B] CA [auth B] DA [auth B] EA [auth B] | SULFATE ION O4 S QAOWNCQODCNURD-UHFFFAOYSA-L |  | ||
| GOL Download:Ideal Coordinates CCD File | F [auth A] IA [auth C] K [auth A] MA [auth C] VA [auth D] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| DMS Download:Ideal Coordinates CCD File | H [auth A] I [auth A] M [auth A] R [auth B] SA [auth D] | DIMETHYL SULFOXIDE C2 H6 O S IAZDPXIOMUYVGZ-UHFFFAOYSA-N |  | ||
| ACY Download:Ideal Coordinates CCD File | L [auth A], LA [auth C] | ACETIC ACID C2 H4 O2 QTBSBXVTEAMEQO-UHFFFAOYSA-N |  | ||
| CMO Download:Ideal Coordinates CCD File | J [auth A], KA [auth C], RA [auth D], W [auth B] | CARBON MONOXIDE C O UGFAIRIUMAVXCW-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 92.178 | α = 90 |
| b = 92.178 | β = 90 |
| c = 142.973 | γ = 120 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| xia2 | data scaling |
| xia2 | data reduction |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Other government | -- |