Cryo-EM structure of Human Nucleosome collected by EPU on Glacios at 3.3 Angstrom resolution
Jia, L., Ruben, E.A., Olsen, S.K., Wasmuth, E.V.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Histone H3.2 | 97 | Homo sapiens | Mutation(s): 0 Gene Names: H3C15, HIST2H3A, H3C14, H3F2, H3FM, HIST2H3C, H3C13, HIST2H3D | 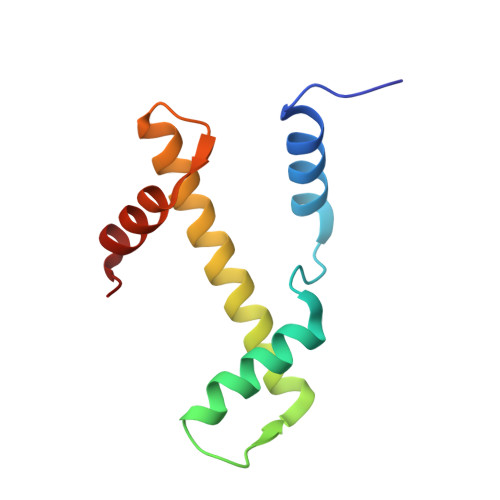 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q71DI3 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q71DI3 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Histone H4 | 83 | Homo sapiens | Mutation(s): 0 |  | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A0A9J8D176 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Histone H2A type 1 | 103 | Homo sapiens | Mutation(s): 0 Gene Names: | 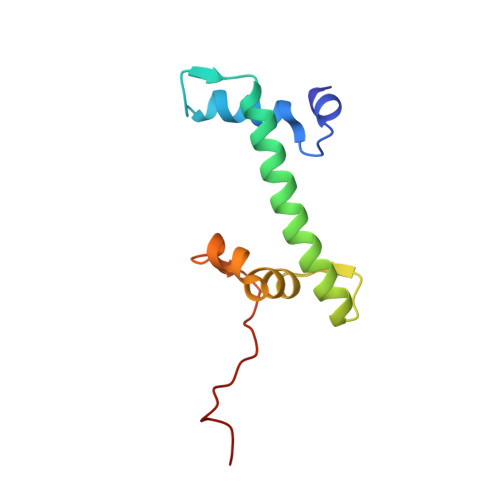 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P0C0S8 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P0C0S8 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 4 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Histone H2B type 1-J | 95 | Homo sapiens | Mutation(s): 0 Gene Names: H2BC11, H2BFR, HIST1H2BJ | 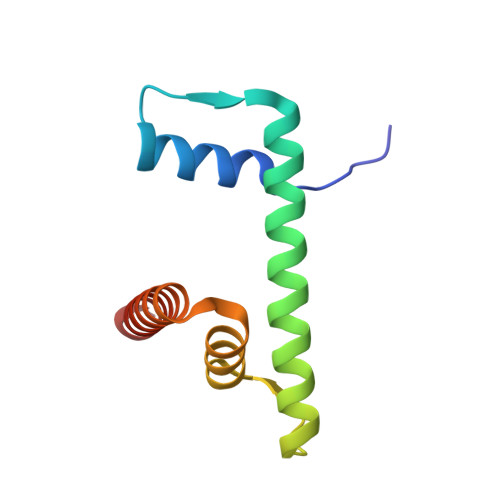 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P06899 GTEx: ENSG00000124635 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P06899 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 5 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Histone H3.2 | 98 | Homo sapiens | Mutation(s): 0 Gene Names: H3C15, HIST2H3A, H3C14, H3F2, H3FM, HIST2H3C, H3C13, HIST2H3D | 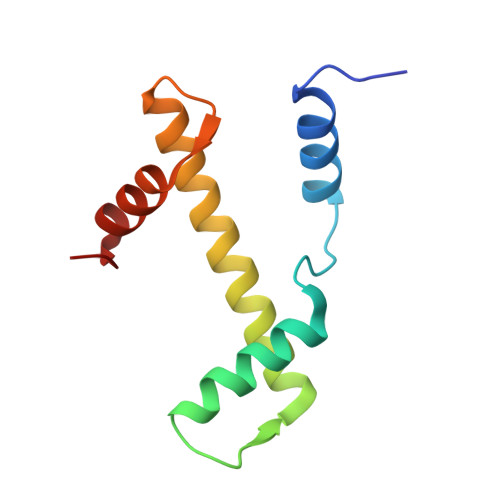 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q71DI3 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q71DI3 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 6 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Histone H2A type 1-B/E | 105 | Homo sapiens | Mutation(s): 0 Gene Names: H2AC4, H2AFM, HIST1H2AB, H2AC8, H2AFA, HIST1H2AE |  | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P04908 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P04908 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 7 | ||||
| Molecule | Chains | Length | Organism | Image |
|---|---|---|---|---|
| DNA (145-MER) | 145 | Homo sapiens |  | |
Sequence AnnotationsExpand | ||||
Reference Sequence | ||||
Entity ID: 8 | ||||
| Molecule | Chains | Length | Organism | Image |
|---|---|---|---|---|
| DNA (145-MER) | 145 | Homo sapiens |  | |
Sequence AnnotationsExpand | ||||
Reference Sequence | ||||
| Task | Software Package | Version |
|---|---|---|
| MODEL REFINEMENT | PHENIX | |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) | United States | -- |