Crystal structure of Aedes aegypti Dopachrome Conversion Enzyme.
Guo, Y., Zhang, L., Wang, S., Guo, D., Deng, J., Li, J., Han, Q.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Dopachrome conversion enzyme | 463 | Aedes aegypti | Mutation(s): 0 | 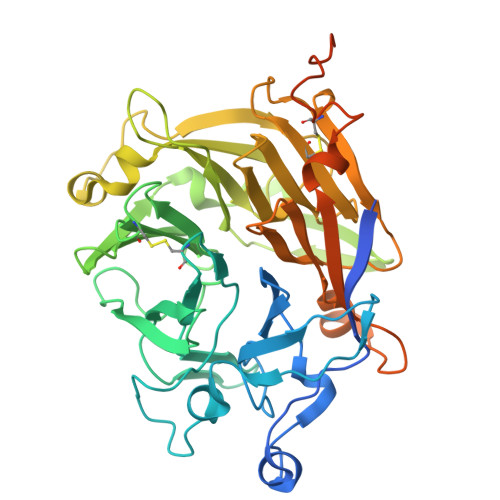 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q8T4S2 | ||||
Glycosylation | |||||
| Glycosylation Sites: 1 | |||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Length | 2D Diagram | Glycosylation | D Interactions |
| beta-D-mannopyranose-(1-3)-[alpha-L-fucopyranose-(1-6)]2-acetamido-2-deoxy-beta-D-glucopyranose | E | 3 |  | N-Glycosylation | |
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Length | 2D Diagram | Glycosylation | D Interactions |
| beta-D-mannopyranose-(1-3)-[alpha-L-fucopyranose-(1-6)]2-acetamido-2-deoxy-alpha-D-glucopyranose | F | 3 |  | N/A | |
Entity ID: 4 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Length | 2D Diagram | Glycosylation | D Interactions |
| beta-D-mannopyranose-(1-4)-[alpha-L-fucopyranose-(1-6)]2-acetamido-2-deoxy-alpha-D-glucopyranose | G | 3 |  | N/A | |
Entity ID: 7 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Length | 2D Diagram | Glycosylation | D Interactions |
| beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose | J | 2 |  | N-Glycosylation | |
Glycosylation Resources | |||||
GlyTouCan: G90333CG GlyCosmos: G90333CG GlyGen: G90333CG | |||||
Entity ID: 8 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Length | 2D Diagram | Glycosylation | D Interactions |
| beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-alpha-D-glucopyranose | K | 2 |  | N/A | |
Glycosylation Resources | |||||
GlyTouCan: G96721TP GlyCosmos: G96721TP GlyGen: G96721TP | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| ACY Download:Ideal Coordinates CCD File | AA [auth C] CA [auth D] DA [auth D] EA [auth D] FA [auth D] | ACETIC ACID C2 H4 O2 QTBSBXVTEAMEQO-UHFFFAOYSA-N |  | ||
| CA Download:Ideal Coordinates CCD File | BA [auth D], M [auth A], U [auth B], Y [auth C] | CALCIUM ION Ca BHPQYMZQTOCNFJ-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 88.76 | α = 90 |
| b = 105.073 | β = 109.97 |
| c = 101.404 | γ = 90 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| SCALEPACK | data scaling |
| HKL-2000 | data reduction |
| MOLREP | phasing |
| PDB_EXTRACT | data extraction |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Institute Of Allergy and Infectious Diseases (NIH/NIAID) | United States | AI044399 |
| National Natural Science Foundation of China (NSFC) | China | U22A20363 |