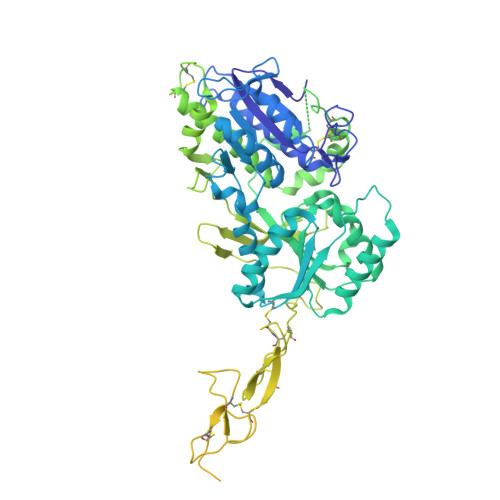
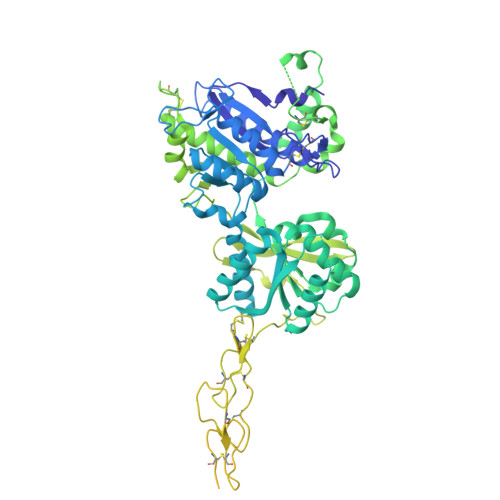
Structure and activation mechanism of human sweet taste receptor.
Wang, H., Chen, X., Dai, Y., Pidathala, S., Niu, Y., Zhao, C., Li, S., Wang, L., Lee, C.H.(2025) Cell Res 35: 775-778
- PubMed: 40754567 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41422-025-01156-x
- Primary Citation Related Structures:
9OPW, 9OPX, 9OPY, 9OPZ, 9OQ0, 9OQ1, 9OQ2, 9OQ3, 9OQ4, 9OQ5, 9OQ6 - Department of Structural Biology, St. Jude Children's Research Hospital, Memphis, TN, USA.
Organizational Affiliation: