Structure of Shigella bacteriophage Sf11
Subramanian, S., Dover, J.A., Parent, K.N.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Major capsid protein gp39 | 356 | Shigella phage Sf11 SMD-2017 | Mutation(s): 0 |  | |
UniProt | |||||
Find proteins for A0A291AXD4 (Shigella phage Sf11 SMD-2017) Explore A0A291AXD4 Go to UniProtKB: A0A291AXD4 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A0A291AXD4 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Putative capsid decoration protein gp40 | 155 | Shigella phage Sf11 SMD-2017 | Mutation(s): 0 | 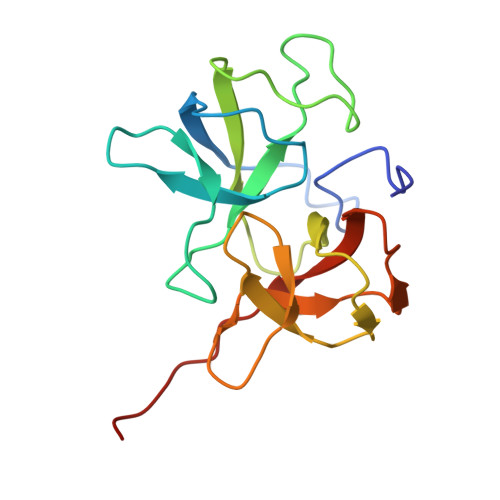 | |
UniProt | |||||
Find proteins for A0A291AXD6 (Shigella phage Sf11 SMD-2017) Explore A0A291AXD6 Go to UniProtKB: A0A291AXD6 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A0A291AXD6 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Task | Software Package | Version |
|---|---|---|
| MODEL REFINEMENT | PHENIX | 1.20.1_4487: |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) | United States | GM116789 |
| National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) | United States | GM140803 |
| National Science Foundation (NSF, United States) | United States | 1750125 |