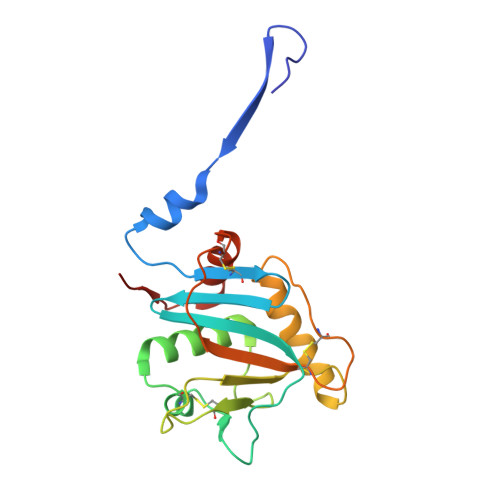
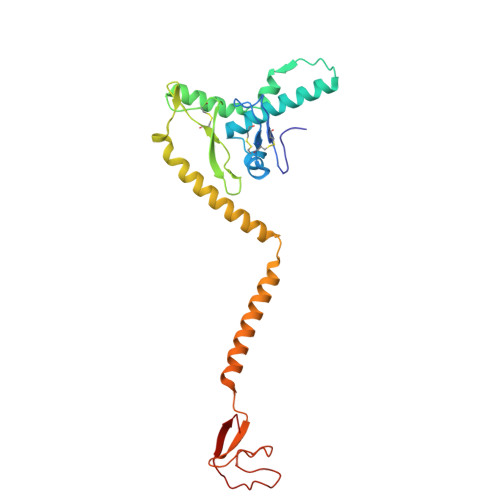
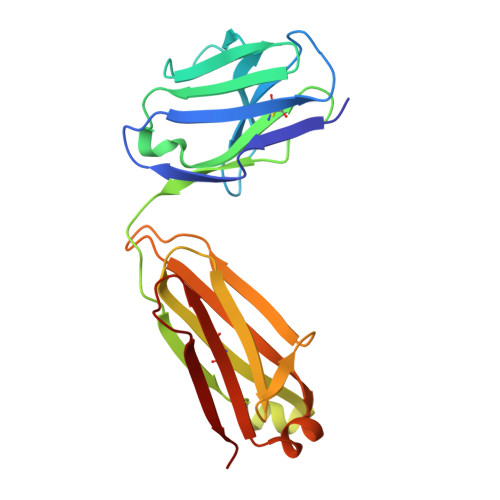
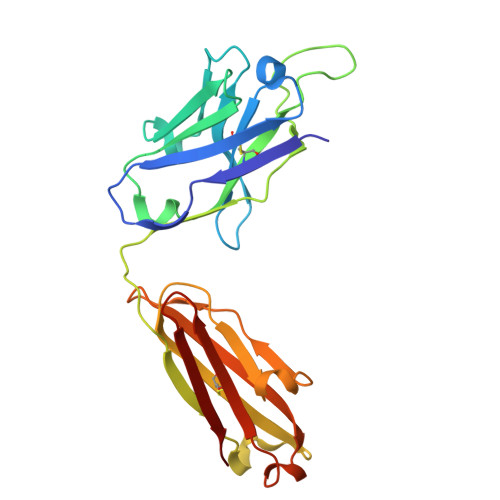
In situ insights into antibody-mediated neutralization of a pre-fusion Junin virus glycoprotein complex.
Taylor, L.J., Sawaya, M.R., Westover, J.B., Wang, C., Jimenez, F., Munoz, A.J., Whitelegge, J., Gowen, B.B., Helguera, G.F., Castells-Graells, R., Rodriguez, J.A.(2025) Cell Rep 44: 115971-115971
- PubMed: 40632652 Search on PubMed
- DOI: https://doi.org/10.1016/j.celrep.2025.115971
- Primary Citation Related Structures:
9MEW, 9N0D - PubMed Abstract:
A transmembrane glycoprotein complex (GPC) decorates the Junin mammarenavirus (JUNV) that causes New World hemorrhagic fevers. We leveraged single-particle cryoelectron microscopy (cryo-EM) to image the full-length JUNV GPC directly on pseudotyped virus (PV) membranes and bound by two JUNV-neutralizing antibodies: Candid#1 vaccine-elicited CR1-28 and J199, a potent therapeutic against Argentine hemorrhagic fever (AHF). The 3.8 Å resolution in situ structures of the antibody-neutralized, 3-fold symmetric JUNV GPC reveal its ectodomain architecture, signal peptide-bound transmembrane region, zinc-binding luminal domain, and post-translational modifications. JUNV-GPC sequence variants highlight the functional importance of the signal peptide transmembrane helix register for virus infection and attenuating Candid#1-associated variants. Overlapping CR1-28 and J199 epitopes suggest a common receptor-blocking mechanism for JUNV neutralization, while a J199-induced, symmetric GPC reorientation may further drive its potent inhibition of JUNV lethality in mice, compared to receptor blockade alone. This underscores the utility of in situ insights into GPC function and neutralization.
- Department of Chemistry and Biochemistry, UCLA-DOE Institute for Genomics and Proteomics, STROBE, NSF Science and Technology Center, University of California, California, Los Angeles (UCLA), Los Angeles, CA 90095, USA. Electronic address: lilyjanetaylor@g.ucla.edu.
Organizational Affiliation: