Structure of the complex of LGR4 with Norrin (2:2)
Yong, G., Hu, F.Z., Qiao, H.R.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| MB52 | A, D [auth B] | 556 | Camelus dromedarius | Mutation(s): 0 | 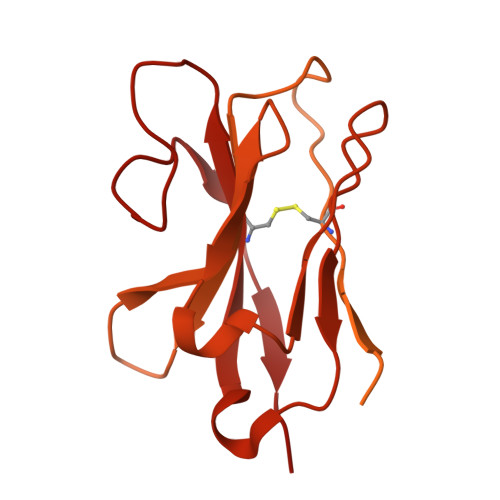 |
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Norrin,Immunoglobulin gamma-1 heavy chain | B [auth E], C [auth F] | 409 | Homo sapiens | Mutation(s): 0 Gene Names: NDP, EVR2 | 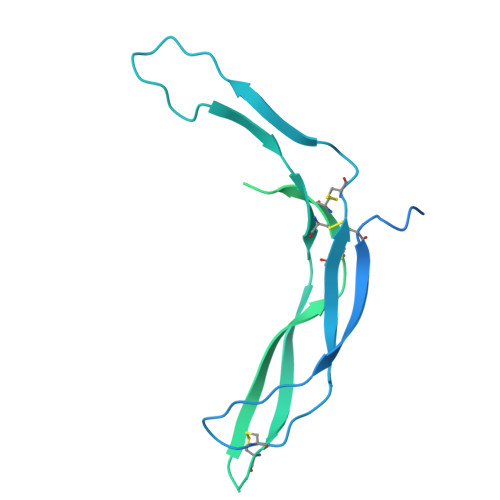 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q00604 GTEx: ENSG00000124479 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q00604 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Leucine-rich repeat-containing G-protein coupled receptor 4 | E [auth C], F [auth D] | 954 | Homo sapiens | Mutation(s): 0 Gene Names: LGR4, GPR48 | 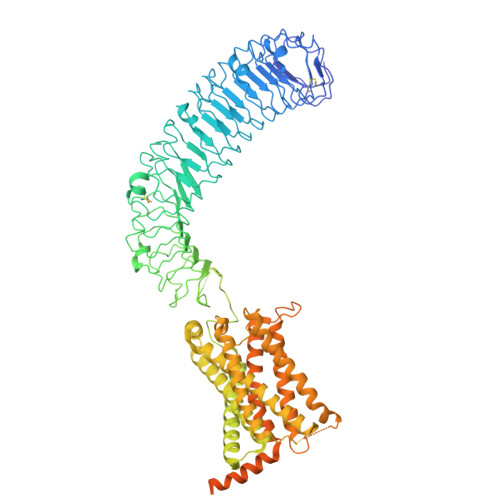 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q9BXB1 GTEx: ENSG00000205213 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9BXB1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Task | Software Package | Version |
|---|---|---|
| MODEL REFINEMENT | PHENIX | |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Natural Science Foundation of China (NSFC) | China | -- |