A cooperative binding mechanism steers the recruitment of peripheral subunits in the E. coli pyruvate dehydrogenase complex
Bothe, S.N., Racunica, D., Zajec Hudnik, T., Zdanowicz, R., Bothe, A., Giese, C., Glockshuber, R.To be published.
Experimental Data Snapshot
Starting Model: in silico
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Dihydrolipoyllysine-residue acetyltransferase component of pyruvate dehydrogenase complex | 66 | Escherichia coli | Mutation(s): 0 Gene Names: aceF, b0115, JW0111 EC: 2.3.1.12 | 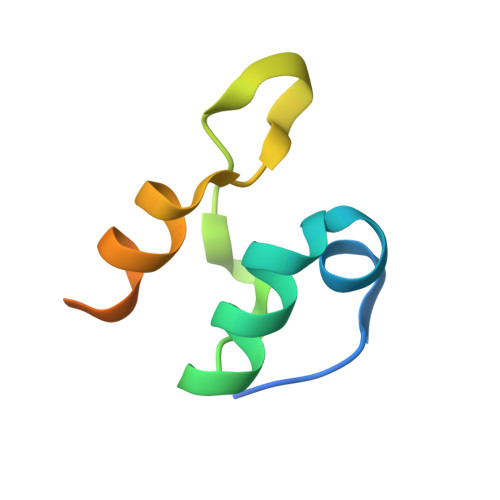 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P06959 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Dihydrolipoyl dehydrogenase | 473 | Escherichia coli | Mutation(s): 0 Gene Names: lpdA, lpd, b0116, JW0112 EC: 1.8.1.4 | 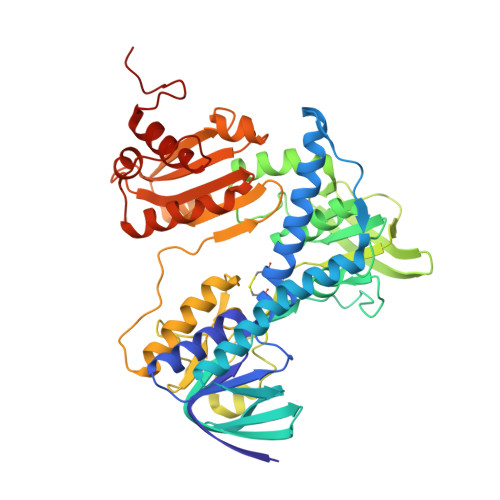 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P0A9P0 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| FAD (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | D [auth B], E [auth C] | FLAVIN-ADENINE DINUCLEOTIDE C27 H33 N9 O15 P2 VWWQXMAJTJZDQX-UYBVJOGSSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 60.703 | α = 90 |
| b = 88.353 | β = 90 |
| c = 200.687 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| PHENIX | refinement |
| XDS | data reduction |
| Aimless | data scaling |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Swiss National Science Foundation | Switzerland | -- |