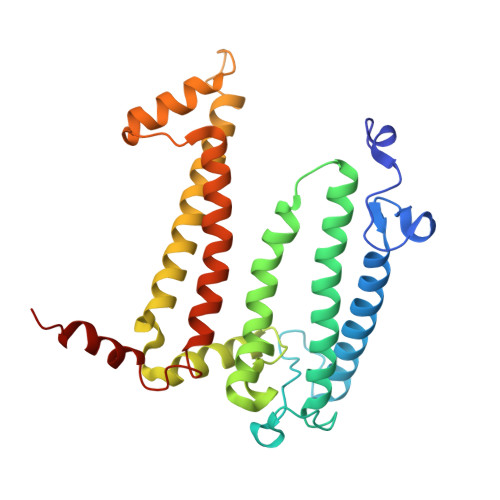
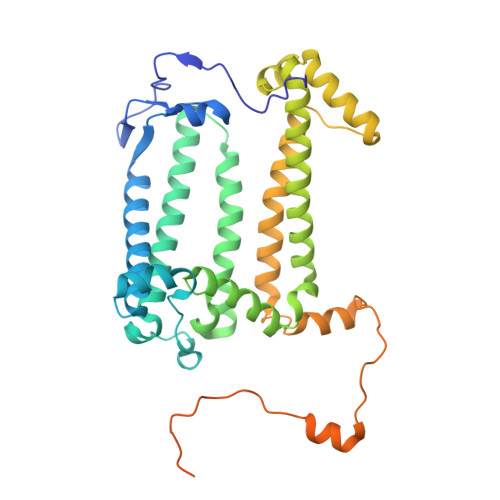
Two solutions for efficient light-harvesting in phototrophic Gemmatimonadota.
Gardiner, A.T., Jin, Y., Bina, D., Joosten, M., Kaftan, D., Mujakic, I., Gardian, Z., Castro-Hartmann, P., Qian, P., Koblizek, M.(2026) mSystems 11: e0109425-e0109425
- PubMed: 41342568 Search on PubMed
- DOI: https://doi.org/10.1128/msystems.01094-25
- Primary Citation Related Structures:
9H19, 9H22 - PubMed Abstract:
Phototrophic Gemmatimonadota represent a unique group of phototrophic bacteria that acquired a complete set of photosynthetic genes via horizontal gene transfer and later evolved independently. Gemmatimonas ( Gem. ) phototrophica contains photosynthetic complexes with two concentric light-harvesting antenna rings that absorb at 816 and 868 nm, allowing it to better exploit the light conditions found deeper in the water column. The closely related species Gem. groenlandica , with highly similar photosynthetic genes, harvests infrared light using a single 860 nm absorption band. The cryo-electron microscopy structure of the Gem. groenlandica photosynthetic complex reveals that the outer antenna lacks monomeric bacteriochlorophylls, resulting in a smaller optical antenna cross-section. The Gem. groenlandica spectrum is red-shifted relative to Gem. phototrophica due to the formation of a H-bond enabled by a different rotamer conformation of αTrp 31 in the outer ring. This H-bond forms with a neighboring bacteriochlorophyll and increases the intra-dimer exciton coupling, affecting the exciton localization probability within the rings and increasing exciton cooperativity between the complexes. The functional consequences of the spectral shift, caused solely by a subtle conformational change of a single residue, represent a novel mechanism in which phototrophic organisms adjust their antennae for particular light conditions and enable Gem. groenlandica to grow higher in the water column where more photons are available.IMPORTANCEThe photoheterotrophic species of the phylum Gemmatimonadota employ unique photosynthetic complexes with two concentric antenna rings around a central reaction center. In contrast to other phototrophic species, these organisms have not evolved any regulatory systems to control the expression of their photosynthetic apparatus under different light conditions. Despite the overall similarity, the complexes present in Gemmatimonas phototrophica and Gemmatimonas groenlandica have different absorption properties in the near-infrared region of the spectrum that make them more suitable for low or medium light, respectively. The main difference in absorption depends on the conformation of a single tryptophan residue that can form an H-bond with a neighboring bacteriochlorophyll. The presence or absence of this H-bond affects how the protein scaffold interacts with the bacteriochlorophylls, which in turn determines how light energy is transferred within and between the photosynthetic complexes.
- Institute of Microbiology of the Czech Academy of Sciences, Třeboň, Czech Republic.
Organizational Affiliation: