Entire structure of ancestral alphaproteobacterial complex I reveals the mechanism to prevent its deactivation
Yaikhomba, M., Hirst, J., Croll, T.I., Spikes, T.E.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-quinone oxidoreductase subunit A | 121 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1 |  | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B498 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-quinone oxidoreductase subunit B | 175 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1 |  | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B497 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-quinone oxidoreductase subunit C | 208 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1 |  | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B496 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 4 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-quinone oxidoreductase subunit D | 412 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1 |  | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B495 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 5 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH dehydrogenase subunit E | 239 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 1.6.5.3 |  | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B494 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 6 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-quinone oxidoreductase subunit F | 431 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1 |  | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B491 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 7 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-quinone oxidoreductase | 674 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1 |  | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B489 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 8 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-quinone oxidoreductase subunit H | 345 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1 |  | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B487 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 9 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-quinone oxidoreductase subunit I | 163 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1 |  | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B486 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 10 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-quinone oxidoreductase subunit J | 199 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1 |  | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B483 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 11 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-quinone oxidoreductase subunit K | 101 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1 |  | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B482 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 12 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH dehydrogenase subunit L | 703 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 1.6.5.3 |  | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B481 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 13 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH dehydrogenase subunit M | 513 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 1.6.5.3 |  | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B480 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 14 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-quinone oxidoreductase subunit N | 499 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1 |  | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B479 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 15 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NAD-dependent epimerase/dehydratase | O [auth P] | 330 | Paracoccus denitrificans PD1222 | Mutation(s): 0 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1AZB0 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 16 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| ETC complex I subunit conserved region | P [auth Q] | 103 | Paracoccus denitrificans PD1222 | Mutation(s): 0 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B1M0 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 17 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Zinc finger CHCC-type domain-containing protein | Q [auth R] | 62 | Paracoccus denitrificans PD1222 | Mutation(s): 0 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B357 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 18 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Protein-L-isoaspartate O-methyltransferase | R [auth Z] | 217 | Paracoccus denitrificans PD1222 | Mutation(s): 0 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B5L6 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 19 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Cytochrome b | S [auth a], V [auth d] | 440 | Paracoccus denitrificans PD1222 | Mutation(s): 0 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B4F3 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 20 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Cytochrome c1 | T [auth b], W [auth e] | 450 | Paracoccus denitrificans PD1222 | Mutation(s): 0 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B4F4 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 21 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Ubiquinol-cytochrome c reductase iron-sulfur subunit | U [auth c], X [auth f] | 195 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1.8 | 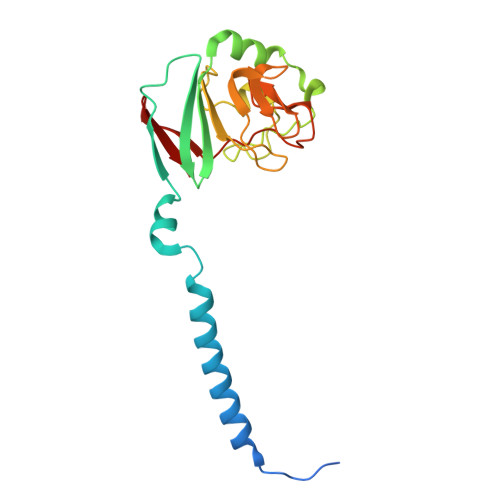 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B4F2 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 22 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Cytochrome c oxidase subunit 1 | CA [auth k], Y [auth g] | 558 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1.9 | 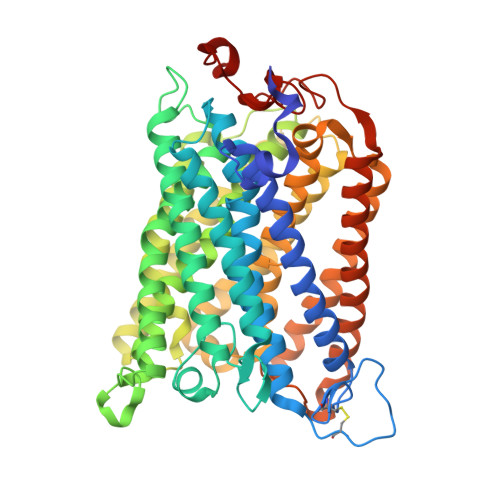 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B3D7 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 23 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Cytochrome c oxidase subunit 2 | DA [auth l], Z [auth h] | 298 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1.9 | 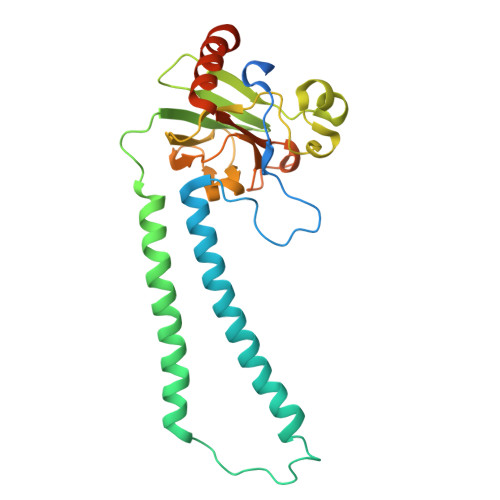 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1BA41 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 24 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| cytochrome-c oxidase | AA [auth i], EA [auth m] | 274 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1.9 | 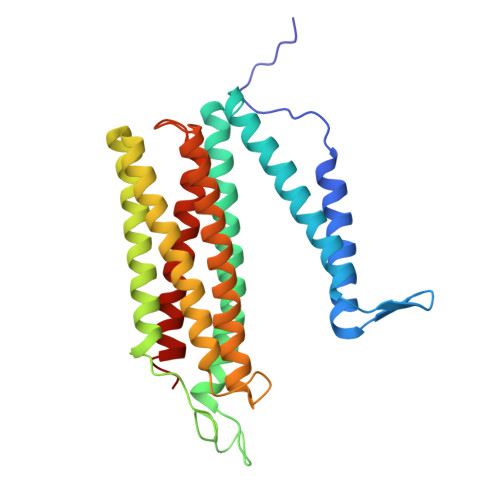 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1BA37 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 25 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Aa3 type cytochrome c oxidase subunit IV | BA [auth j], FA [auth n] | 66 | Paracoccus denitrificans PD1222 | Mutation(s): 0 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1AZ52 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 26 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Cytochrome c, class I | GA [auth o], HA [auth p] | 176 | Paracoccus denitrificans PD1222 | Mutation(s): 0 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B311 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 27 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH:ubiquinone oxidoreductase 17.2 kD subunit | IA [auth q] | 124 | Paracoccus denitrificans PD1222 | Mutation(s): 0 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B1H8 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 20 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| CDL Download:Ideal Coordinates CCD File | TD [auth d] | CARDIOLIPIN C81 H156 O17 P2 XVTUQDWPJJBEHJ-KZCWQMDCSA-L |  | ||
| T7X Download:Ideal Coordinates CCD File | HF [auth g], JB [auth J] | Phosphatidylinositol C47 H83 O13 P KRTOMQDUKGRFDJ-TWUHCGEESA-N |  | ||
| U10 Download:Ideal Coordinates CCD File | CD [auth a], DD [auth a], NA [auth B], WD [auth d], YD [auth d] | UBIQUINONE-10 C59 H90 O4 ACTIUHUUMQJHFO-UPTCCGCDSA-N |  | ||
| HEA (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | CF [auth g], PG [auth k], QG [auth k], YE [auth g] | HEME-A C49 H56 Fe N4 O6 ZGGYGTCPXNDTRV-PRYGPKJJSA-L |  | ||
| P5S Download:Ideal Coordinates CCD File | ED [auth a], FB [auth I], WB [auth L] | O-[(R)-{[(2R)-2,3-bis(octadecanoyloxy)propyl]oxy}(hydroxy)phosphoryl]-L-serine C42 H82 N O10 P TZCPCKNHXULUIY-RGULYWFUSA-N |  | ||
| PC1 Download:Ideal Coordinates CCD File | AG [auth i] AH [auth l] BE [auth d] DG [auth i] DH [auth m] | 1,2-DIACYL-SN-GLYCERO-3-PHOSPHOCHOLINE C44 H88 N O8 P NRJAVPSFFCBXDT-HUESYALOSA-N |  | ||
| PGT Download:Ideal Coordinates CCD File | AC [auth M] BB [auth H] FD [auth a] IB [auth J] KA [auth A] | (1S)-2-{[{[(2R)-2,3-DIHYDROXYPROPYL]OXY}(HYDROXY)PHOSPHORYL]OXY}-1-[(PALMITOYLOXY)METHYL]ETHYL STEARATE C40 H79 O10 P KBPVYRBBONZJHF-AMAPPZPBSA-N |  | ||
| 3PE Download:Ideal Coordinates CCD File | AB [auth H] AD [auth a] BG [auth i] CB [auth H] CE [auth d] | 1,2-Distearoyl-sn-glycerophosphoethanolamine C41 H82 N O8 P LVNGJLRDBYCPGB-LDLOPFEMSA-N |  | ||
| 3PH Download:Ideal Coordinates CCD File | AE [auth d] BC [auth M] BD [auth a] BH [auth m] CC [auth M] | 1,2-DIACYL-GLYCEROL-3-SN-PHOSPHATE C39 H77 O8 P YFWHNAWEOZTIPI-DIPNUNPCSA-N |  | ||
| HEC (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | KD [auth b], ME [auth e] | HEME C C34 H34 Fe N4 O4 HXQIYSLZKNYNMH-LJNAALQVSA-N |  | ||
| HEM (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | GD [auth a], GE [auth d], WC [auth a], ZD [auth d] | PROTOPORPHYRIN IX CONTAINING FE C34 H32 Fe N4 O4 KABFMIBPWCXCRK-RGGAHWMASA-L |  | ||
| DU0 Download:Ideal Coordinates CCD File | CH [auth m] DE [auth d] EE [auth d] FC [auth M] FG [auth i] | 2-[2-[(1~{S},2~{S},4~{S},5'~{R},6~{R},7~{S},8~{R},9~{S},12~{S},13~{R},16~{S})-5',7,9,13-tetramethylspiro[5-oxapentacyclo[10.8.0.0^{2,9}.0^{4,8}.0^{13,18}]icos-18-ene-6,2'-oxane]-16-yl]oxyethyl]propane-1,3-diol C32 H52 O5 GFDJQXOBWHMOSQ-LEZUHYJESA-N |  | ||
| FMN (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | SA [auth F] | FLAVIN MONONUCLEOTIDE C17 H21 N4 O9 P FVTCRASFADXXNN-SCRDCRAPSA-N |  | ||
| SF4 Download:Ideal Coordinates CCD File | GB [auth I] HB [auth I] MA [auth B] RA [auth F] UA [auth G] | IRON/SULFUR CLUSTER Fe4 S4 LJBDFODJNLIPKO-UHFFFAOYSA-N |  | ||
| FES Download:Ideal Coordinates CCD File | QA [auth E], QD [auth c], RE [auth f], TA [auth G] | FE2/S2 (INORGANIC) CLUSTER Fe2 S2 NIXDOXVAJZFRNF-UHFFFAOYSA-N |  | ||
| CUA Download:Ideal Coordinates CCD File | OF [auth h], YG [auth l] | DINUCLEAR COPPER ION Cu2 ALKZAGKDWUSJED-UHFFFAOYSA-N |  | ||
| ZN Download:Ideal Coordinates CCD File | CG [auth i], IH [auth m], RC [auth R] | ZINC ION Zn PTFCDOFLOPIGGS-UHFFFAOYSA-N |  | ||
| CU Download:Ideal Coordinates CCD File | BF [auth g], VG [auth k] | COPPER (II) ION Cu JPVYNHNXODAKFH-UHFFFAOYSA-N |  | ||
| MN Download:Ideal Coordinates CCD File | GF [auth g], WG [auth k] | MANGANESE (II) ION Mn WAEMQWOKJMHJLA-UHFFFAOYSA-N |  | ||
| CA Download:Ideal Coordinates CCD File | AF [auth g], JE [auth e], LD [auth b], NG [auth k], PA [auth D] | CALCIUM ION Ca BHPQYMZQTOCNFJ-UHFFFAOYSA-N |  | ||
| Modified Residues 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| FME Query on FME | A | L-PEPTIDE LINKING | C6 H11 N O3 S |  | MET |
| 2MR Query on 2MR | D | L-PEPTIDE LINKING | C8 H18 N4 O2 |  | ARG |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Medical Research Council (MRC, United Kingdom) | United Kingdom | MC_UU_00015/2 and MC_UU_00028/1 |