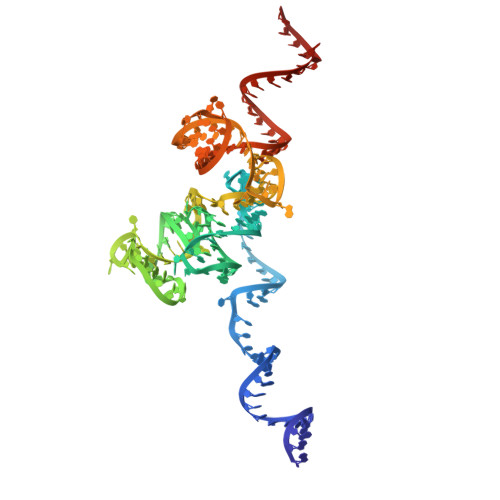
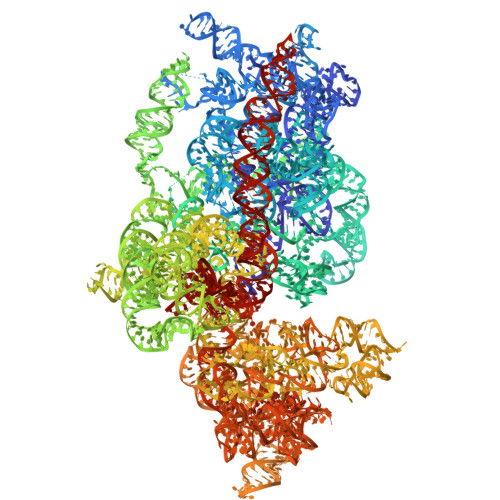
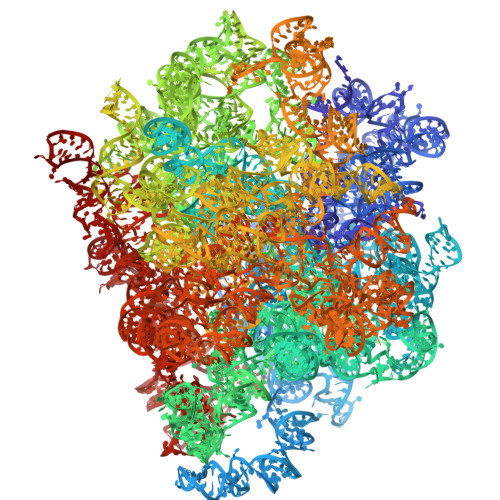
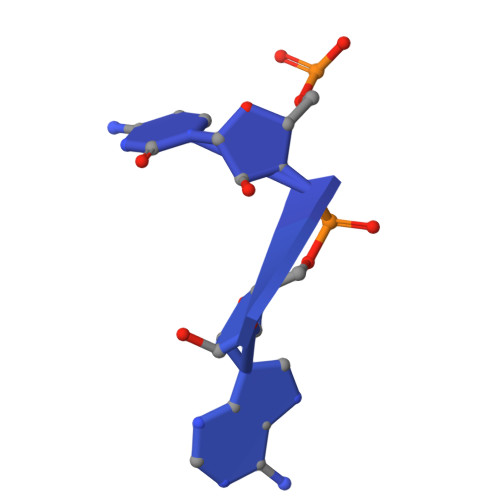
Mechanism of read-through enhancement by aminoglycosides and mefloquine.
Kolosova, O., Zgadzay, Y., Stetsenko, A., Sukhinina, A.P., Atamas, A., Validov, S., Rogachev, A., Usachev, K., Jenner, L., Dmitriev, S.E., Yusupova, G., Guskov, A., Yusupov, M.(2025) Proc Natl Acad Sci U S A 122: e2420261122-e2420261122
- PubMed: 40273100 Search on PubMed
- DOI: https://doi.org/10.1073/pnas.2420261122
- Primary Citation Related Structures:
9G1Z, 9G30, 9G6J - PubMed Abstract:
Nonsense mutations are associated with numerous and diverse pathologies, yet effective treatment strategies remain elusive. A promising approach to combat these conditions involves the use of aminoglycosides, particularly in combination with stop-codon read-through enhancers, for developing drugs that can rescue the production of full-length proteins. Using X-ray crystallography and single-particle cryo-EM, we obtained structures of the eukaryotic ribosome in complexes with several aminoglycosides (geneticin G418, paromomycin, and hygromycin B) and the antimalarial drug mefloquine (MFQ), which has also been identified as a read-through enhancer. Our study reveals a binding site of MFQ, which holds significant promise for the development of therapies targeting premature termination codon-related genetic and oncological diseases. The results underscore the crucial role of the bridge B7b/c in mediating the effects of MFQ on subunit rotation dynamics. Through a comprehensive analysis of the interactions between the drugs and the eukaryotic ribosome, we propose a unifying hypothesis for read-through enhancement by small molecules, highlighting the role of decoding center rearrangements and intersubunit rotation dynamics.
- Department of Integrated Structural Biology, Institute of Genetics and Molecular and Cellular Biology, University of Strasbourg, Illkirch 67400, France.
Organizational Affiliation: