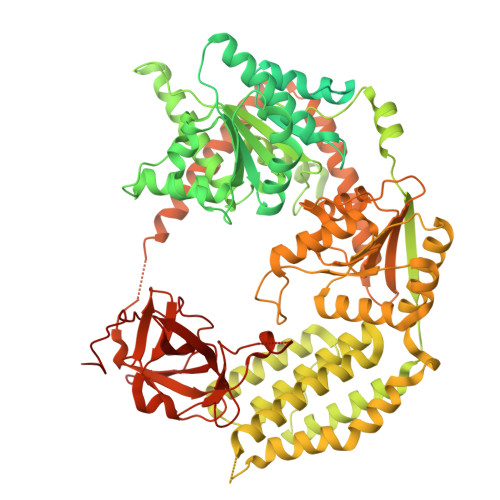
Molecular basis of autoimmune disease protection by MDA5 variants.
Singh, R., Joiner, J.D., Herrero Del Valle, A., Zwaagstra, M., Jobe, I., Ferguson, B.J., van Kuppeveld, F.J.M., Modis, Y.(2025) Cell Rep 44: 115754-115754
- PubMed: 40450684 Search on PubMed
- DOI: https://doi.org/10.1016/j.celrep.2025.115754
- Primary Citation Related Structures:
9F0J, 9F1U, 9F20, 9F2L, 9F2W, 9F3P - PubMed Abstract:
MDA5 recognizes double-stranded RNA (dsRNA) from viruses and retroelements. Cooperative filament formation and ATP-dependent proofreading confer MDA5 with the necessary sensitivity and specificity for dsRNA. Many MDA5 genetic variants are associated with protection from autoimmune disease while increasing the risk of infection and chronic inflammation. How these variants affect RNA sensing remains unclear. Here, we determine the consequences of autoimmune-protective variants on the molecular structure and activities of MDA5. Rare variants E627 ∗ and I923V reduce the interferon response to picornavirus infection. E627 ∗ does not bind RNA. I923V is ATPase hyperactive, causing premature dissociation from dsRNA. Cryoelectron microscopy (cryo-EM) structures of MDA5 I923V bound to dsRNA at different stages of ATP hydrolysis reveal smaller RNA binding interfaces, leading to excessive proofreading activity. Variants R843H and T946A, which are genetically linked and cause mild phenotypes, did not affect cytokine induction, suggesting an indirect disease mechanism. In conclusion, autoimmune-protective MDA5 variants dampen MDA5-dependent signaling via multiple mechanisms.
- Molecular Immunity Unit, Department of Medicine, University of Cambridge, MRC Laboratory of Molecular Biology, Cambridge CB2 0QH, UK; Cambridge Institute of Therapeutic Immunology & Infectious Disease (CITIID), Department of Medicine, University of Cambridge, Cambridge CB2 0AW, UK; Department of Pathology, University of Cambridge, Cambridge CB2 1QP, UK.
Organizational Affiliation: