Photoswitching fluorescence via a novel Trp-flap mechanism
Bingham, C., Ehyaei, N., Maity, S., Cantrell, J., Gwasdacus, J., Sheng, W., Vasileiou, C., Borhan, B., Geiger, J.H.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Retinol-binding protein 2 | 133 | Homo sapiens | Mutation(s): 9 Gene Names: RBP2, CRBP2 | 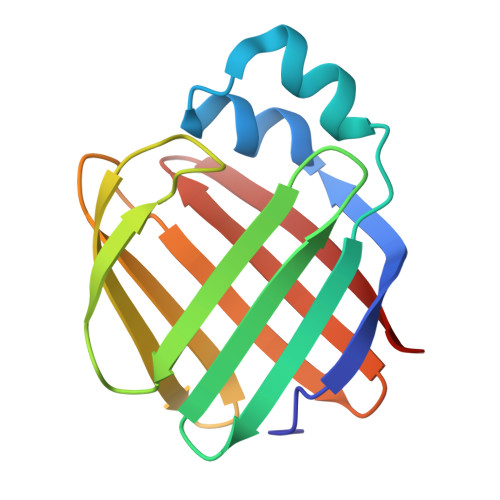 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P50120 GTEx: ENSG00000114113 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P50120 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| ZFJ (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | C [auth A], G [auth B] | (4aP)-N,N-diethyl-9,9-dimethyl-7-[(1E)-prop-1-en-1-yl]-9H-fluoren-2-amine C22 H27 N UMARKYXLRJLTSK-RMKNXTFCSA-N |  | ||
| GOL Download:Ideal Coordinates CCD File | D [auth A], H [auth B] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| ACY Download:Ideal Coordinates CCD File | E [auth A], F [auth A], I [auth B] | ACETIC ACID C2 H4 O2 QTBSBXVTEAMEQO-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 31.1 | α = 86.11 |
| b = 35.997 | β = 86.5 |
| c = 64.013 | γ = 65.1 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |
| PHENIX | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Institute of Biomedical Imaging and Bioengineering (NIH/NIBIB) | United States | -- |