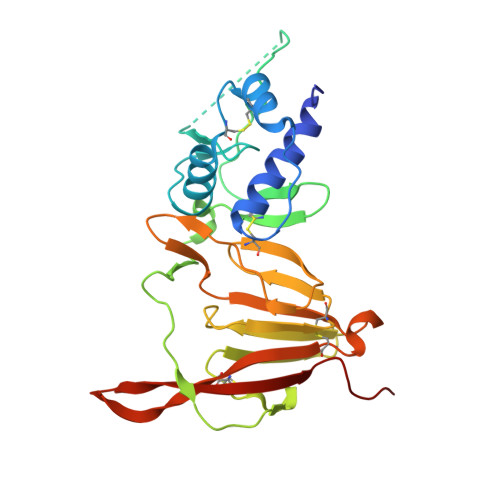
Engineering and structures of Crimean-Congo hemorrhagic fever virus glycoprotein complexes.
McFadden, E., Monticelli, S.R., Wang, A., Ramamohan, A.R., Batchelor, T.G., Kuehne, A.I., Bakken, R.R., Tse, A.L., Chandran, K., Herbert, A.S., McLellan, J.S.(2025) Cell 188: 303
- PubMed: 39701101 Search on PubMed
- DOI: https://doi.org/10.1016/j.cell.2024.11.008
- Primary Citation Related Structures:
9AVF, 9B8J - PubMed Abstract:
Crimean-Congo hemorrhagic fever virus (CCHFV) is a tickborne virus that can cause severe disease in humans with case fatality rates of 10%-40%. Although structures of CCHFV glycoproteins GP38 and Gc have provided insights into viral entry and defined epitopes of neutralizing and protective antibodies, the structure of glycoprotein Gn and its interactions with GP38 and Gc have remained elusive. Here, we use structure-guided protein engineering to produce a stabilized GP38-Gn-Gc heterotrimeric glycoprotein complex (GP38-Gn H-DS -Gc). A cryo-electron microscopy (cryo-EM) structure of this complex provides the molecular basis for GP38's association on the viral surface, reveals the structure of Gn, and demonstrates that GP38-Gn restrains the Gc fusion loops in the prefusion conformation, facilitated by an N-linked glycan attached to Gn. Immunization with GP38-Gn H-DS -Gc conferred 40% protection against lethal IbAr10200 challenge in mice. These data define the architecture of a GP38-Gn-Gc protomer and provide a template for structure-guided vaccine antigen development.
- Department of Molecular Biosciences, The University of Texas at Austin, Austin, TX 78712, USA.
Organizational Affiliation: