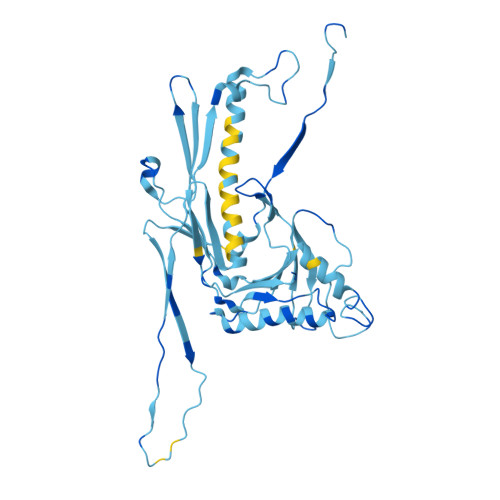
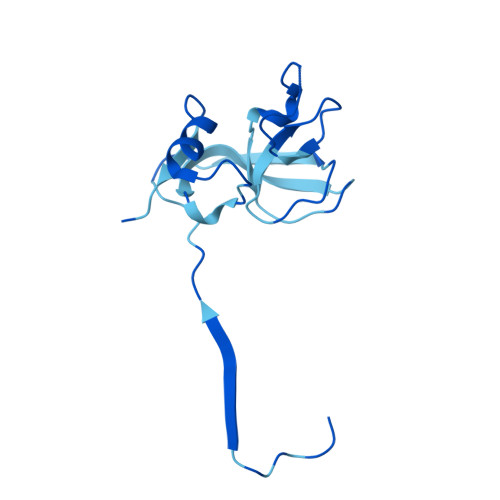
Cryo-EM structure of flagellotropic bacteriophage Chi.
Sonani, R.R., Esteves, N.C., Scharf, B.E., Egelman, E.H.(2024) Structure 32: 856-865.e3
- PubMed: 38614087 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.str.2024.03.011
- Primary Citation Related Structures:
8VHX, 8VJA, 8VJH, 8VJI - PubMed Abstract:
The flagellotropic bacteriophage χ (Chi) infects bacteria via the flagellar filament. Despite years of study, its structural architecture remains partly characterized. Through cryo-EM, we unveil χ's nearly complete structure, encompassing capsid, neck, tail, and tail tip. While the capsid and tail resemble phage YSD1, the neck and tail tip reveal new proteins and their arrangement. The neck shows a unique conformation of the tail tube protein, forming a socket-like structure for attachment to the neck. The tail tip comprises four proteins, including distal tail protein (DTP), two baseplate hub proteins (BH1P and BH2P), and tail tip assembly protein (TAP) exhibiting minimal organization compared to other siphophages. Deviating from the consensus in other siphophages, DTP in χ forms a trimeric assembly, reducing tail symmetry from 6-fold to 3-fold at the tip. These findings illuminate the previously unexplored structural organization of χ's neck and tail tip.
- Department of Biochemistry and Molecular Genetics, University of Virginia School of Medicine, Charlottesville, VA 22903, USA.
Organizational Affiliation: