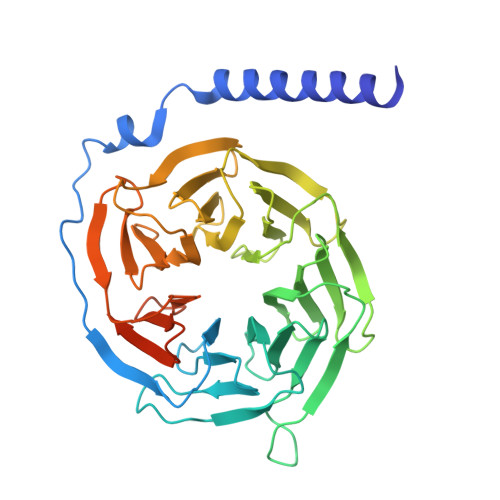
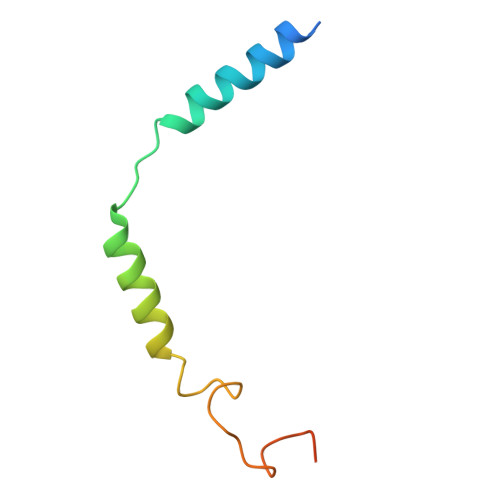
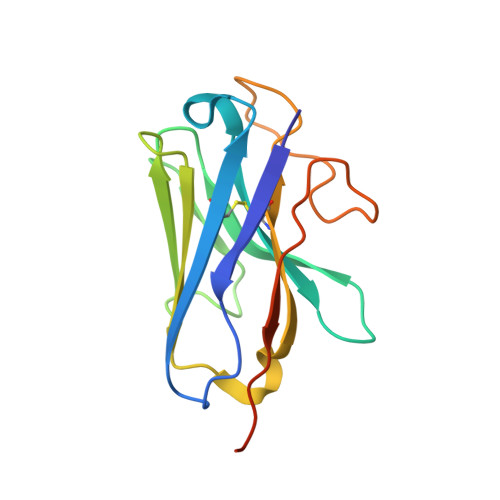
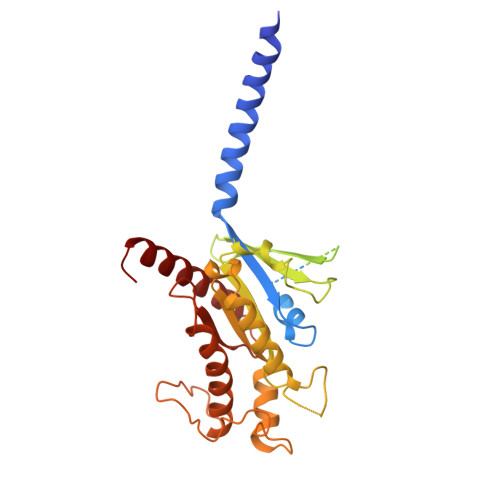
Native mass spectrometry prescreening of G protein-coupled receptor complexes for cryo-EM structure determination.
Kim, D., Liu, W., Viner, R., Cherezov, V.(2024) Structure 32: 2206-2219.e4
- PubMed: 39471802 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.str.2024.10.004
- Primary Citation Related Structures:
8VHF - PubMed Abstract:
G protein-coupled receptors (GPCRs) are essential transmembrane proteins playing key roles in human health and disease. Understanding their atomic-level molecular structure and conformational states is imperative for advancing drug development. Recent breakthroughs in single-particle cryogenic electron microscopy (cryo-EM) have propelled the structural biology of GPCRs into a new era. Nevertheless, the preparation of suitable GPCR samples and their complexes for cryo-EM analysis remains challenging due to their poor stability and highly dynamic nature. Here, we present our online buffer exchange-native MS method combined with Direct Mass Technology (OBE-nMS+DMT) which facilitates high-throughput analysis and guides sample preparation. We applied this method to optimize the GPR119-G s complex sample prior to cryo-EM analysis, leading to a 3.51 Å resolution structure from only 396 movies collected on a 200 kV Glacios. This study suggests that the OBE-nMS+DMT method emerges as a powerful tool for prescreening sample conditions in cryo-EM studies of GPCRs and other membrane protein complexes.
- Bridge Institute, Michelson Center for Convergent Bioscience, University of Southern California, Los Angeles, CA 90089, USA; Department of Chemistry, University of Southern California, Los Angeles, CA 90089, USA.
Organizational Affiliation: