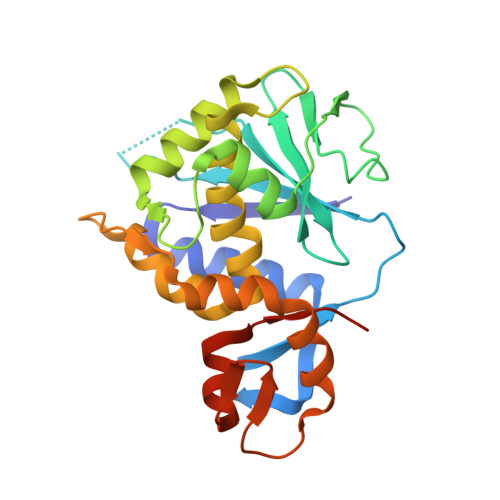
Structure-Function Analysis of the A1 Subunit of Shiga Toxin 2 with Peptides That Target the P-Stalk Binding Site and Inhibit Activity.
Li, X.P., Rudolph, M.J., Chen, Y., Tumer, N.E.(2024) Biochemistry 63: 893-905
- PubMed: 38467020
- DOI: https://doi.org/10.1021/acs.biochem.3c00733
- Primary Citation Related Structures:
8SZ2 - PubMed Abstract:
Shiga toxin 2a (Stx2a) is the virulence factor of Escherichia coli (STEC), which is associated with hemolytic uremic syndrome, the leading cause of pediatric kidney failure. The A1 subunit of Stx2a (Stx2A1) binds to the conserved C-terminal domain (CTD) of the ribosomal P-stalk proteins to remove an adenine from the sarcin-ricin loop (SRL) in the 28S rRNA, inhibiting protein synthesis. There are no antidotes against Stx2a or any other ribosome-inactivating protein (RIP). The structural and functional details of the binding of Stx2A1 to the P-stalk CTD are not known. Here, we carry out a deletion analysis of the conserved P-stalk CTD and show that the last eight amino acids (P8) of the P-stalk proteins are the minimal sequence required for optimal affinity and maximal inhibitory activity against Stx2A1. We determined the first X-ray crystal structure of Stx2A1 alone and in complex with P8 and identified the exact binding site. The C-terminal aspartic acid of the P-stalk CTD serves as an anchor, forming key contacts with the conserved arginine residues at the P-stalk binding pocket of Stx2A1. Although the ricin A subunit (RTA) binds to the P-stalk CTD, the last aspartic acid is more critical for the interaction with Stx2A1, indicating that RIPs differ in their requirements for the P-stalk. These results demonstrate that the catalytic activity of Stx2A1 is inhibited by blocking its interactions with the P-stalk, providing evidence that P-stalk binding is an essential first step in the recruitment of Stx2A1 to the SRL for depurination.
- Department of Plant Biology, Rutgers, The State University of New Jersey, 59 Dudley Road, New Brunswick, New Jersey 08901, United States.
Organizational Affiliation: