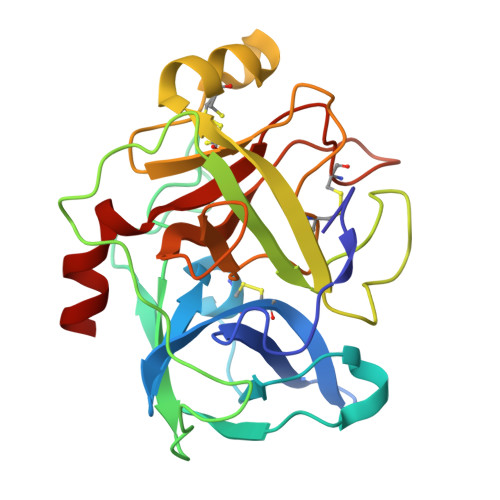
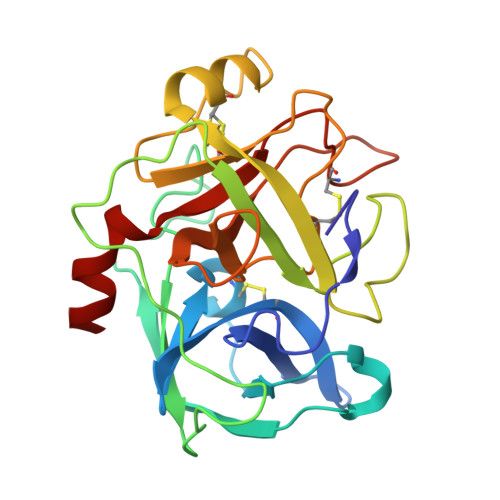
A facultative plasminogen-independent thrombolytic enzyme from Sipunculus nudus.
Tang, M., Ma, G., Xu, C., Yang, H., Lin, H., Bian, C., Hu, C., Lu, M., Chen, L., Jie, W., Yue, Z., Jian, J., Sun, Y., Yan, H., Zhou, J., Zhang, X., Liao, S., Li, Z., Cai, S., Wu, Y., Yang, K., Xiong, Y., Zhao, Y., Lv, Z., Xu, X., Liu, C., Xin, P., Ye, L., Cui, X., Shi, Q., Chen, X., Xu, R.(2025) Nat Commun 16: 3852-3852
- PubMed: 40274794 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-025-58915-y
- Primary Citation Related Structures:
8ZVS, 8ZVX - PubMed Abstract:
Current thrombolytic therapies primarily function by converting plasminogen into plasmin, a process dependent on the fibrin-activator complex. This dependence, coupled with the substantial molecular size of plasmin, constrains its effectiveness in degrading D-dimer and restricts its diffusion within thrombi. Here, we introduce a small facultative plasminogen-independent thrombolytic enzyme, snFPITE, isolated from Sipunculus nudus. Compared to traditional thrombolytic agents, snFPITE does not require plasminogen for thrombolysis, although its presence enhances lytic activity. This enzyme fully degrades cross-linked fibrin without leaving residual nondegradable D-dimer and generates a smaller fibrinolytic-active agent from plasminogen. A series of male rats and mice models further confirm that snFPITE is a safety injectable thrombolytic agent. Mechanistically, snFPITE activates plasminogen and degrades fibrin(ogen) in a multisite cleavage manner. snFPITE is inhibited by plasminogen activator inhibitor 1 and α2-antiplasmin via a competitive inhibition. We further identify 28 snFPITE candidate sequences, of which 10 are confirmed as functional genes.
- Engineering Research Centre of Molecular Medicine of Ministry of Education, Fujian Key Laboratory of Molecular Medicine, Key Laboratory of Precision Medicine and Molecular Diagnosis of Fujian Universities, Xiamen Key Laboratory of Marine and Gene Drugs, School of Biomedical Sciences, Huaqiao University, Xiamen, Fujian, China.
Organizational Affiliation: