Structural basis of ceramide transportation and its regulation by ABCH transporter
Chen, J., Zhou, Y., Yang, Q.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| ABC transporter G family member 23-like Protein | 791 | Tribolium castaneum | Mutation(s): 0 Gene Names: AUGUSTUS-3.0.2_34444, TcasGA2_TC034444 | 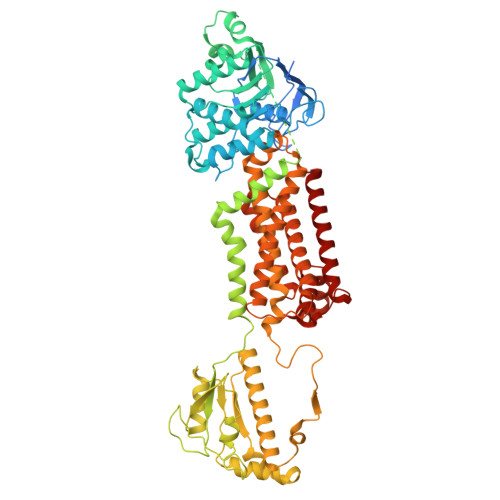 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A0A139WBS7 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| AV0 (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | C [auth A], E [auth B] | Lauryl Maltose Neopentyl Glycol C47 H88 O22 MADJBYLAYPCCOO-VWHTXWAPSA-N |  | ||
| A1D7S (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | D [auth A], F [auth B] | [(2~{R})-1-[2-azanylethoxy(oxidanyl)phosphoryl]oxy-3-[(5~{Z},8~{Z},11~{E},14~{E},17~{Z})-icosa-5,8,11,14,17-pentaenoxy]propan-2-yl] octadecanoate C43 H78 N O7 P BCVRSQNGADAZKS-GZACMAKJSA-N |  | ||
| A1D7T Download:Ideal Coordinates CCD File | G [auth B] | (~{E})-~{N}-[(2~{S},3~{R})-1,3-bis(oxidanyl)octadecan-2-yl]octadec-9-enamide C36 H71 N O3 MJQIARGPQMNBGT-CJDDIQBSSA-N |  | ||
| Task | Software Package | Version |
|---|---|---|
| MODEL REFINEMENT | PHENIX | 1.20.1_4487: |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Natural Science Foundation of China (NSFC) | China | 32161133010 |