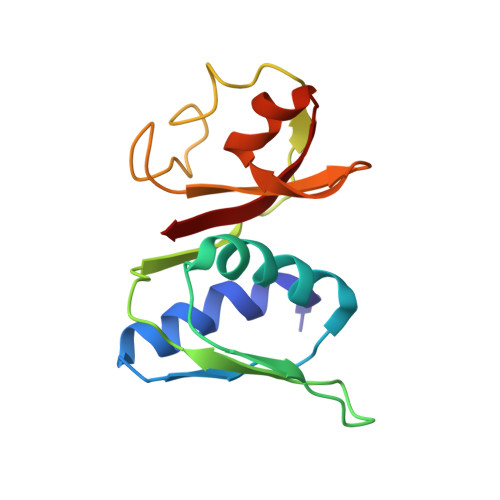
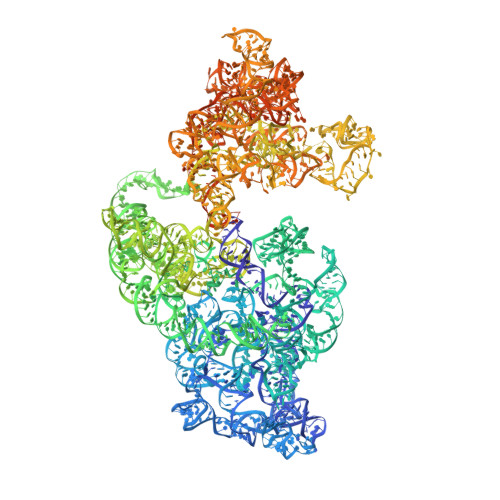
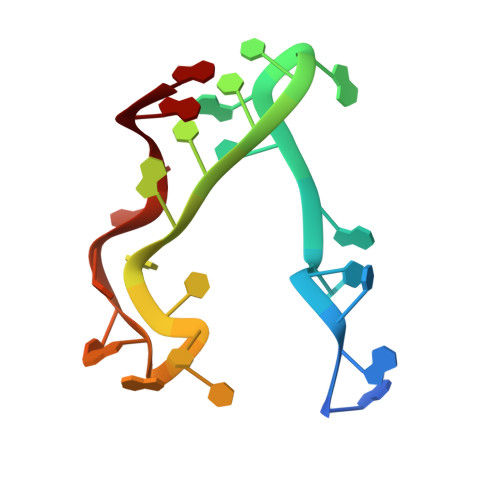
Structural basis of translation inhibition by a valine tRNA-derived fragment.
Wu, Y., Ni, M.T., Wang, Y.H., Wang, C., Hou, H., Zhang, X., Zhou, J.(2024) Life Sci Alliance 7
- PubMed: 38599770 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.26508/lsa.202302488
- Primary Citation Related Structures:
8WKP, 8WQ2, 8WQ4 - PubMed Abstract:
Translational regulation by non-coding RNAs is a mechanism commonly used by cells to fine-tune gene expression. A fragment derived from an archaeal valine tRNA (Val-tRF) has been previously identified to bind the small subunit of the ribosome and inhibit translation in Haloferax volcanii Here, we present three cryo-electron microscopy structures of Val-tRF bound to the small subunit of Sulfolobus acidocaldarius ribosomes at resolutions between 4.02 and 4.53 Å. Within these complexes, Val-tRF was observed to bind to conserved RNA-interacting sites, including the ribosomal decoding center. The binding of Val-tRF destabilizes helices h24, h44, and h45 and the anti-Shine-Dalgarno sequence of 16S rRNA. The binding position of this molecule partially overlaps with the translation initiation factor aIF1A and occludes the mRNA P-site codon. Moreover, we found that the binding of Val-tRF is associated with steric hindrance of the H69 base of 23S rRNA in the large ribosome subunit, thereby preventing 70S assembly. Our data exemplify how tRNA-derived fragments bind to ribosomes and provide new insights into the mechanisms underlying translation inhibition by Val-tRFs.
- Life Sciences Institute, Zhejiang University, Hangzhou, China.
Organizational Affiliation: