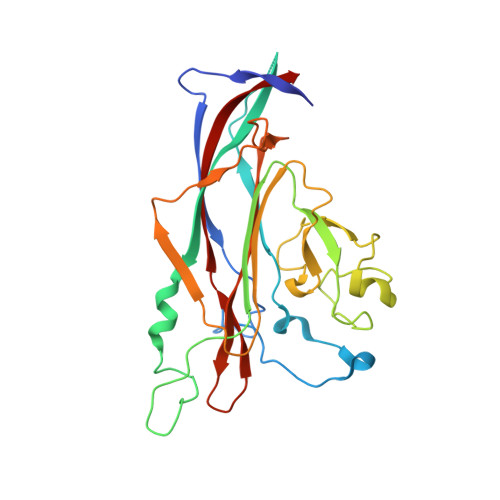
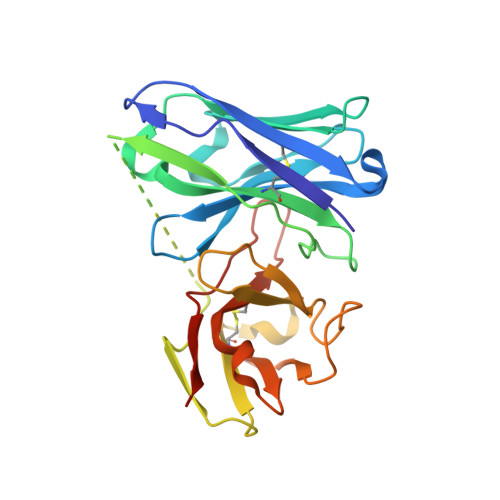
Nonclinical and clinical characterization of MAU868, a novel human-derived monoclonal neutralizing antibody targeting BK polyomavirus VP1.
Abend, J.R., Sathe, A., Wrobel, M.B., Knapp, M., Xu, L., Zhao, L., Kim, P., Desai, S., Nguyen, A., Leber, X.C., Hein, A., Scharenberg, M., Shaul, J., Ornelas, E., Wong, K., Pietzonka, T., Sterling, L.M., Hodges, M.R., Pertel, P., Traggiai, E., Patick, A.K., Kovacs, S.J.(2024) Am J Transplant 24: 1994-2006
- PubMed: 38996969 Search on PubMed
- DOI: https://doi.org/10.1016/j.ajt.2024.07.002
- Primary Citation Related Structures:
8U5L - PubMed Abstract:
Reactivation of BK polyomavirus (BKPyV) can cause significant kidney and bladder disease in immunocompromised patients. There are currently no effective, BKPyV-specific therapies. MAU868 is a novel, human immunoglobulin (Ig) G1 monoclonal antibody that binds the major capsid protein, VP1, of BKPyV with picomolar affinity, neutralizes infection by the 4 major BKPyV genotypes (EC 50 ranging from 0.009-0.093 μg/mL; EC 90 ranging from 0.102-4.160 μg/mL), and has comparable activity against variants with highly prevalent VP1 polymorphisms. No resistance-associated variants were identified in long-term selection studies, indicating a high in vitro barrier-to-resistance. The high-resolution crystal structure of MAU868 in complex with VP1 pentamer identified 3 key contact residues in VP1 (Y169, R170, and K172). A first-in-human study was conducted to assess the safety, tolerability, and pharmacokinetics of MAU868 following intravenous and subcutaneous administration to healthy adults in a randomized, placebo-controlled, double-blinded, single ascending dose design. MAU868 was safe and well-tolerated. All adverse events were grade 1 and resolved. The pharmacokinetics of MAU868 was typical of a human IgG, with dose-proportional systemic exposure and an elimination half-life ranging between 23 and 30 days. These results demonstrate the potential of MAU868 as a first-in-class therapeutic agent for the treatment or prevention of BKPyV disease.
- Novartis Institutes for BioMedical Research, Infectious Disease Area, Emeryville, California, USA. Electronic address: joabend@gmail.com.
Organizational Affiliation: