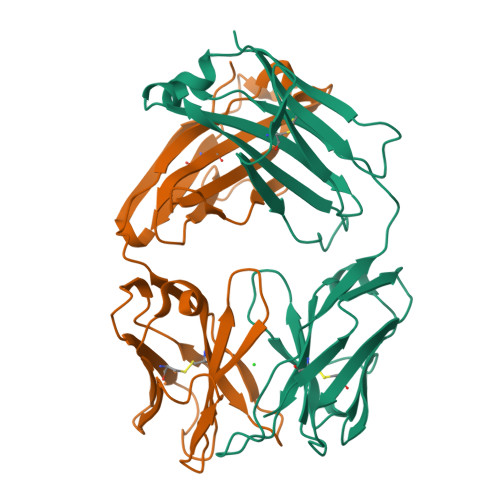
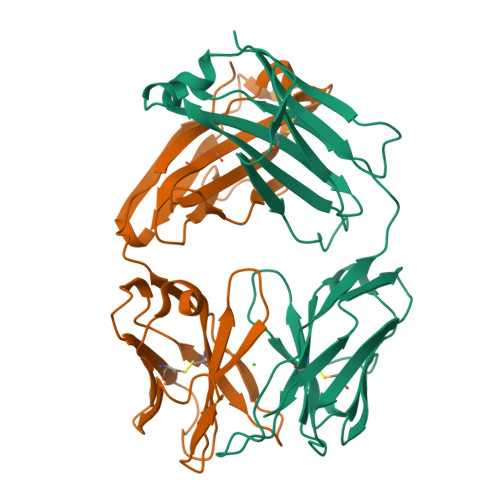
Generation and Study of Antibodies against Two Triangular Trimers Derived from A beta.
Kreutzer, A.G., Malonis, R.J., Parrocha, C.M.T., Tong, K., Guaglianone, G., Nguyen, J.T., Diab, M.N., Lai, J.R., Nowick, J.S.(2024) Pept Sci (Hoboken) 116
- PubMed: 38644932
- DOI: https://doi.org/10.1002/pep2.24333
- Primary Citation of Related Structures:
8T0O - PubMed Abstract:
Monoclonal antibodies (mAbs) that target the P-amyloid peptide (Aβ) are important Alzheimer's disease research tools and are now being used as Alzheimer's disease therapies. Conformation-specific mAbs that target oligomeric and fibrillar Aβ assemblies are of particular interest, as these assemblies are associated with Alzheimer's disease pathogenesis and progression. This paper reports the generation of rabbit mAbs against two different triangular trimers derived from Aβ. These antibodies are the first mAbs generated against Aβ oligomer mimics in which the high-resolution structures of the oligomers are known. We describe the isolation of the mAbs using single B-cell sorting of peripheral blood mononuclear cells (PBMCs) from immunized rabbits, the selectivity of the mAbs for the triangular trimers, the immunoreactivity of the mAbs with aggregated Aβ 42 , and the immunoreactivity of the mAbs in brain tissue from the 5xFAD Alzheimer's disease mouse model. The characterization of these mAbs against structurally defined trimers derived from Aβ enhances understanding of antibody-amyloid recognition and may benefit the development of diagnostics and immunotherapies in Alzheimer's disease.
Organizational Affiliation:
Department of Chemistry, University of California Irvine, Irvine, CA 92697.