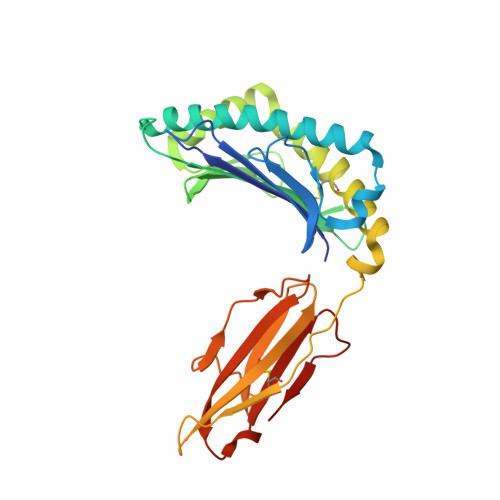
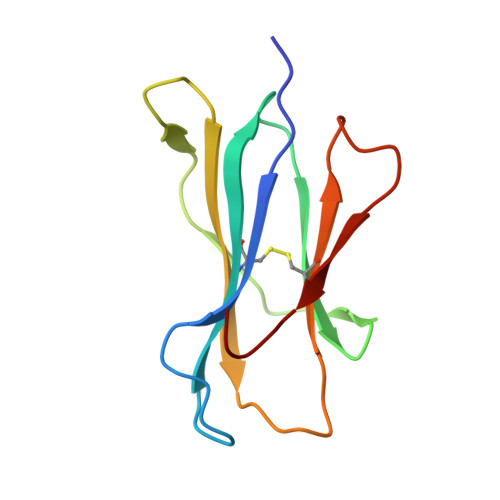
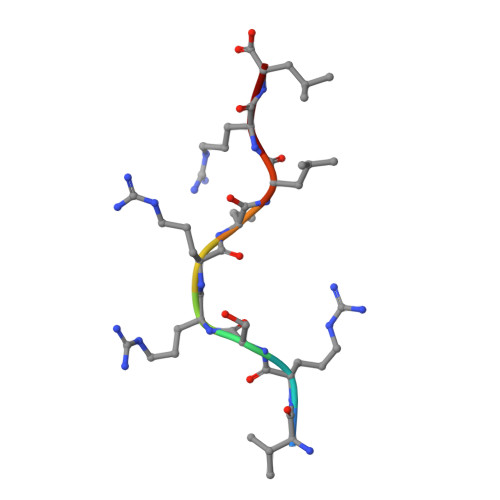
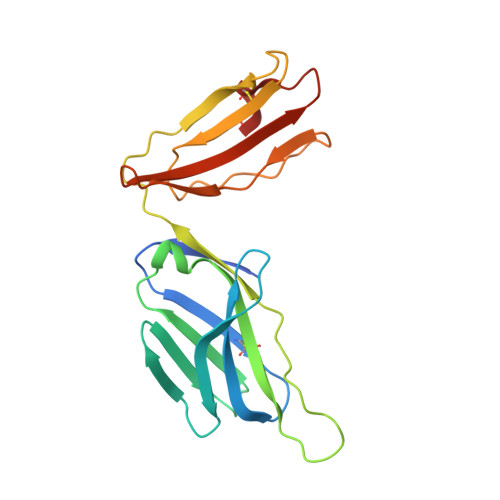
Complimentary electrostatics dominate T-cell receptor binding to a psoriasis-associated peptide antigen presented by human leukocyte antigen C∗06:02.
Anand, S., Littler, D.R., Mobbs, J.I., Braun, A., Baker, D.G., Tennant, L., Purcell, A.W., Vivian, J.P., Rossjohn, J.(2023) J Biological Chem 299: 104930-104930
- PubMed: 37330172 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.jbc.2023.104930
- Primary Citation Related Structures:
8SHI - PubMed Abstract:
Psoriasis is a chronic skin disease characterized by hyperproliferative epidermal lesions infiltrated by autoreactive T cells. Individuals expressing the human leukocyte antigen (HLA) C∗06:02 allele are at highest risk for developing psoriasis. An autoreactive T cell clone (termed Vα3S1/Vβ13S1) isolated from psoriatic plaques is selective for HLA-C∗06:02, presenting a peptide derived from the melanocyte-specific autoantigen ADAMTSL5 (VRSRRCLRL). Here we determine the crystal structure of this psoriatic TCR-HLA-C∗06:02 ADAMTSL5 complex with a stabilized peptide. Docking of the TCR involves an extensive complementary charge network formed between negatively charged TCR residues interleaving with exposed arginine residues from the self-peptide and the HLA-C∗06:02 α1 helix. We probed these interactions through mutagenesis and activation assays. The charged interface spans the polymorphic region of the C1/C2 HLA group. Notably the peptide-binding groove of HLA-C∗06:02 appears exquisitely suited for presenting highly charged Arg-rich epitopes recognized by this acidic psoriatic TCR. Overall, we provide a structural basis for understanding the engagement of melanocyte antigen-presenting cells by a TCR implicated in psoriasis while simultaneously expanding our knowledge of how TCRs engage HLA-C.
- Infection and Immunity Program & Department of Biochemistry and Molecular Biology, Biomedicine Discovery Institute, Monash University, Clayton, Victoria, Australia.
Organizational Affiliation: