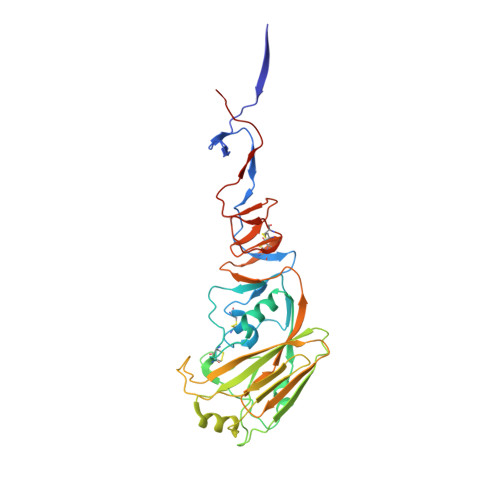
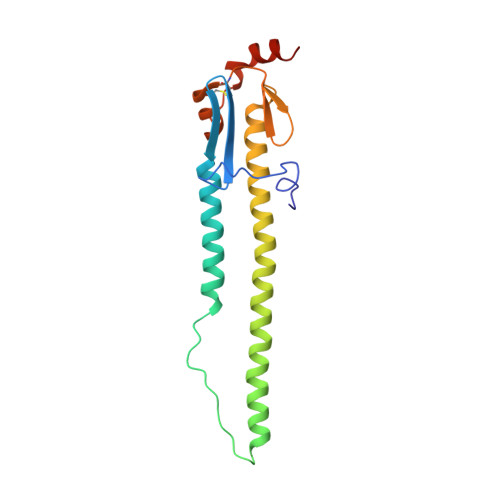
Ultrapotent influenza hemagglutinin fusion inhibitors developed through SuFEx-enabled high-throughput medicinal chemistry.
Kitamura, S., Lin, T.H., Lee, C.D., Takamura, A., Kadam, R.U., Zhang, D., Zhu, X., Dada, L., Nagai, E., Yu, W., Yao, Y., Sharpless, K.B., Wilson, I.A., Wolan, D.W.(2024) Proc Natl Acad Sci U S A 121: e2310677121-e2310677121
- PubMed: 38753503 Search on PubMed
- DOI: https://doi.org/10.1073/pnas.2310677121
- Primary Citation Related Structures:
8SD2, 8SD4, 8VQL, 8VQM, 8VQN, 8VQQ - PubMed Abstract:
Seasonal and pandemic-associated influenza strains cause highly contagious viral respiratory infections that can lead to severe illness and excess mortality. Here, we report on the optimization of our small-molecule inhibitor F0045(S) targeting the influenza hemagglutinin (HA) stem with our Sulfur-Fluoride Exchange (SuFEx) click chemistry-based high-throughput medicinal chemistry (HTMC) strategy. A combination of SuFEx- and amide-based lead molecule diversification and structure-guided design led to identification and validation of ultrapotent influenza fusion inhibitors with subnanomolar EC 50 cellular antiviral activity against several influenza A group 1 strains. X-ray structures of six of these compounds with HA indicate that the appended moieties occupy additional pockets on the HA surface and increase the binding interaction, where the accumulation of several polar interactions also contributes to the improved affinity. The compounds here represent the most potent HA small-molecule inhibitors to date. Our divergent HTMC platform is therefore a powerful, rapid, and cost-effective approach to develop bioactive chemical probes and drug-like candidates against viral targets.
- Department of Integrative Structural and Computational Biology, The Scripps Research Institute, La Jolla, CA 92037.
Organizational Affiliation: