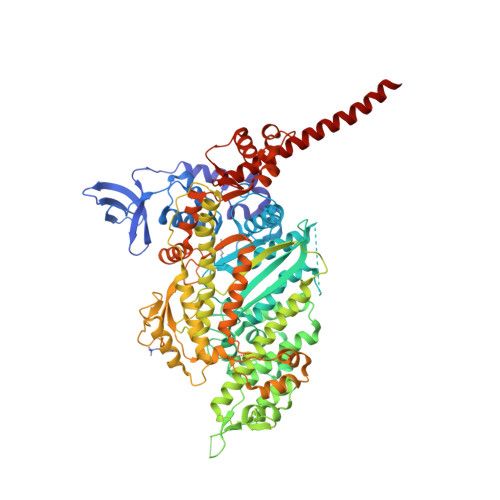
Omecamtiv mecarbil and Mavacamten target the same myosin pocket despite antagonistic effects in heart contraction.
Auguin, D., Robert-Paganin, J., Rety, S., Kikuti, C., David, A., Theumer, G., Schmidt, A.W., Knolker, H.J., Houdusse, A.(2023) bioRxiv
- PubMed: 38014327 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1101/2023.11.15.567213
- Primary Citation Related Structures:
8QYP, 8QYQ, 8QYR, 8QYU - PubMed Abstract:
Inherited cardiomyopathies are amongst the most common cardiac diseases worldwide, leading in the late-stage to heart failure and death. The most promising treatments against these diseases are small-molecules directly modulating the force produced by β-cardiac myosin, the molecular motor driving heart contraction. Two of these molecules that produce antagonistic effects on cardiac contractility have completed clinical phase 3 trials: the activator Omecamtiv mecarbil and the inhibitor Mavacamten . In this work, we reveal by X-ray crystallography that both drugs target the same pocket and stabilize a pre-stroke structural state, with only few local differences. All atoms molecular dynamics simulations reveal how these molecules can have antagonistic impact on the allostery of the motor by comparing β-cardiac myosin in the apo form or bound to Omecamtiv mecarbil or Mavacamten . Altogether, our results provide the framework for rational drug development for the purpose of personalized medicine.
- Structural Motility, UMR 144 CNRS/Curie Institute, PSL Research University, 26 rue d'Ulm, 75258 Paris cedex 05, France.
Organizational Affiliation: