Cryo-EM structure of the complete inner kinetochore of the budding yeast point centromere.
Dendooven, T., Zhang, Z., Yang, J., McLaughlin, S.H., Schwab, J., Scheres, S.H.W., Yatskevich, S., Barford, D.(2023) Sci Adv 9: eadg7480-eadg7480
- PubMed: 37506202 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1126/sciadv.adg7480
- Primary Citation Related Structures:
8OVW, 8OVX, 8OW0, 8OW1 - PubMed Abstract:
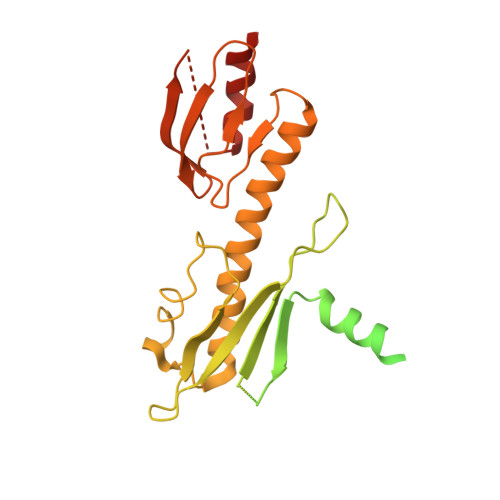
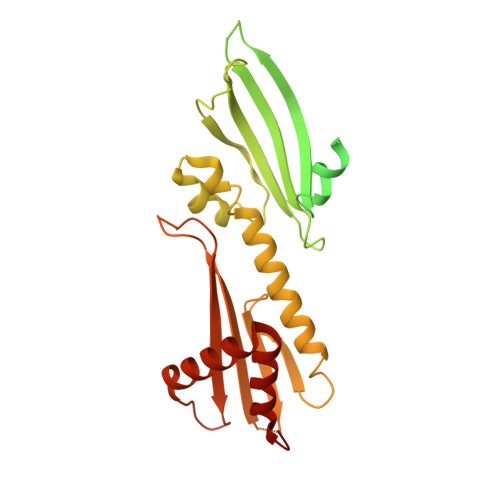
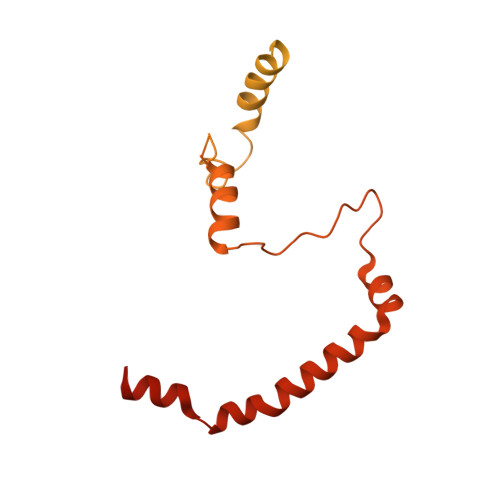
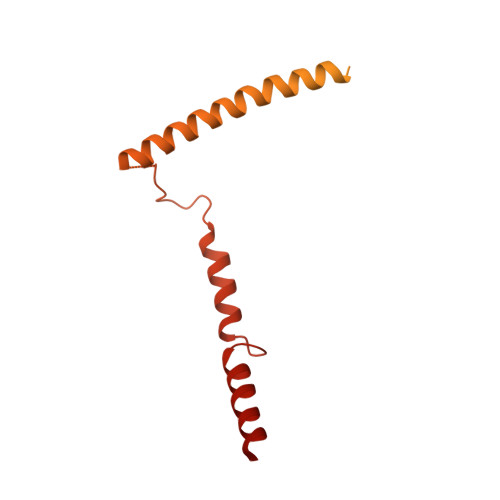
The point centromere of budding yeast specifies assembly of the large kinetochore complex to mediate chromatid segregation. Kinetochores comprise the centromere-associated inner kinetochore (CCAN) complex and the microtubule-binding outer kinetochore KNL1-MIS12-NDC80 (KMN) network. The budding yeast inner kinetochore also contains the DNA binding centromere-binding factor 1 (CBF1) and CBF3 complexes. We determined the cryo-electron microscopy structure of the yeast inner kinetochore assembled onto the centromere-specific centromere protein A nucleosomes (CENP-A Nuc ). This revealed a central CENP-A Nuc with extensively unwrapped DNA ends. These free DNA duplexes bind two CCAN protomers, one of which entraps DNA topologically, positioned on the centromere DNA element I (CDEI) motif by CBF1. The two CCAN protomers are linked through CBF3 forming an arch-like configuration. With a structural mechanism for how CENP-A Nuc can also be linked to KMN involving only CENP-QU, we present a model for inner kinetochore assembly onto a point centromere and how it organizes the outer kinetochore for chromosome attachment to the mitotic spindle.
- MRC Laboratory of Molecular Biology, Francis Crick Avenue, Cambridge CB2 0QH, UK.
Organizational Affiliation: