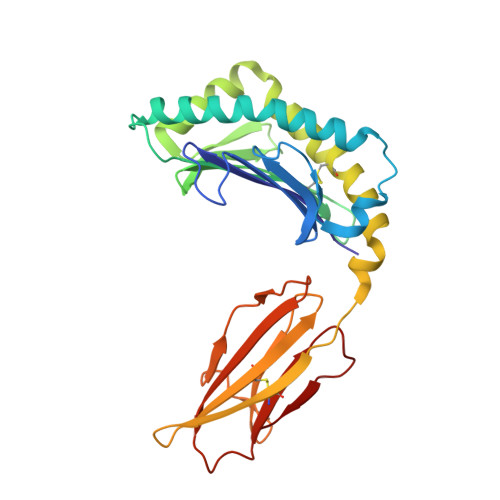
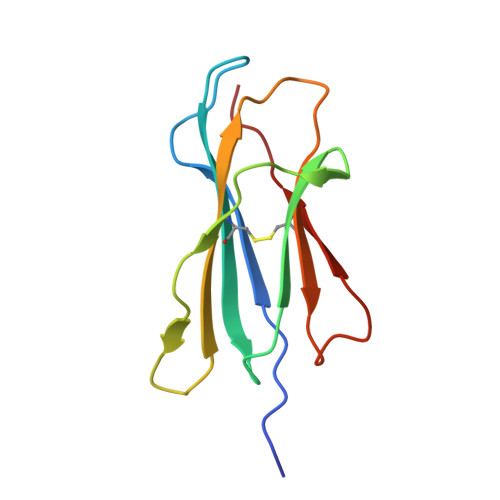
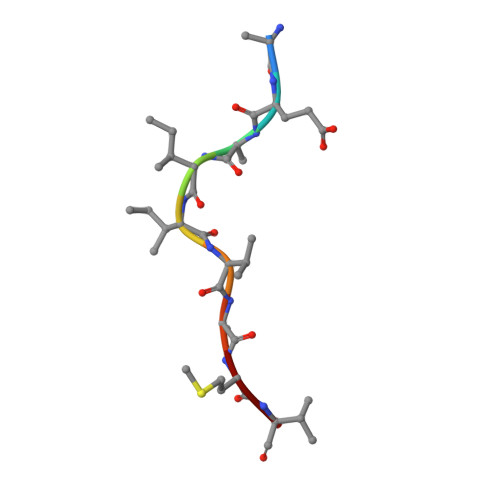
The impact of micropolymorphism in Anpl-UAA on structural stability and peptide presentation.
Tang, Z., Wang, S., Du, L., Hu, D., Chen, X., Zheng, H., Ding, H., Chen, S., Zhang, L., Zhang, N.(2024) Int J Biol Macromol 267: 131665-131665
- PubMed: 38636758 Search on PubMed
- DOI: https://doi.org/10.1016/j.ijbiomac.2024.131665
- Primary Citation Related Structures:
8KB0, 8KB1 - PubMed Abstract:
Micropolymorphism significantly shapes the peptide-binding characteristics of major histocompatibility complex class I (MHC-I) molecules, affecting the host's resistance to pathogens, which is particularly pronounced in avian species displaying the "minimal essential MHC" expression pattern. In this study, we compared two duck MHC-I alleles, Anpl-UAA*77 and Anpl-UAA*78, that exhibit markedly different peptide binding properties despite their high sequence homology. Through mutagenesis experiments and crystallographic analysis of complexes with the influenza virus-derived peptide AEAIIVAMV (AEV9), we identified a critical role for the residue at position 62 in regulating hydrogen-bonding interactions between the peptide backbone and the peptide-binding groove. This modulation affects the characteristics of the B pocket and the stability of the loop region between the 3 10 helix and the α1 helix, leading to significant changes in the structure and stability of the peptide-MHC-I complex (pMHC-I). Moreover, the proportion of different residues at position 62 among Anpl-UAAs may reflect the correlation between pAnpl-UAA stability and duck body temperature. This research not only advances our understanding of the Anpl-UAA structure but also deepens our insight into the impact of MHC-I micropolymorphism on peptide binding.
- National Key Laboratory of Veterinary Public Health Security, Key Laboratory of Animal Epidemiology of the Ministry of Agriculture and Rural Affairs, College of Veterinary Medicine, China Agricultural University, Beijing 100193, China.
Organizational Affiliation: